By targeting three key growth pathways at once, researchers eliminated pancreatic tumors in multiple mouse models and prevented the cancer from returning, a promising step toward overcoming treatment resistance.

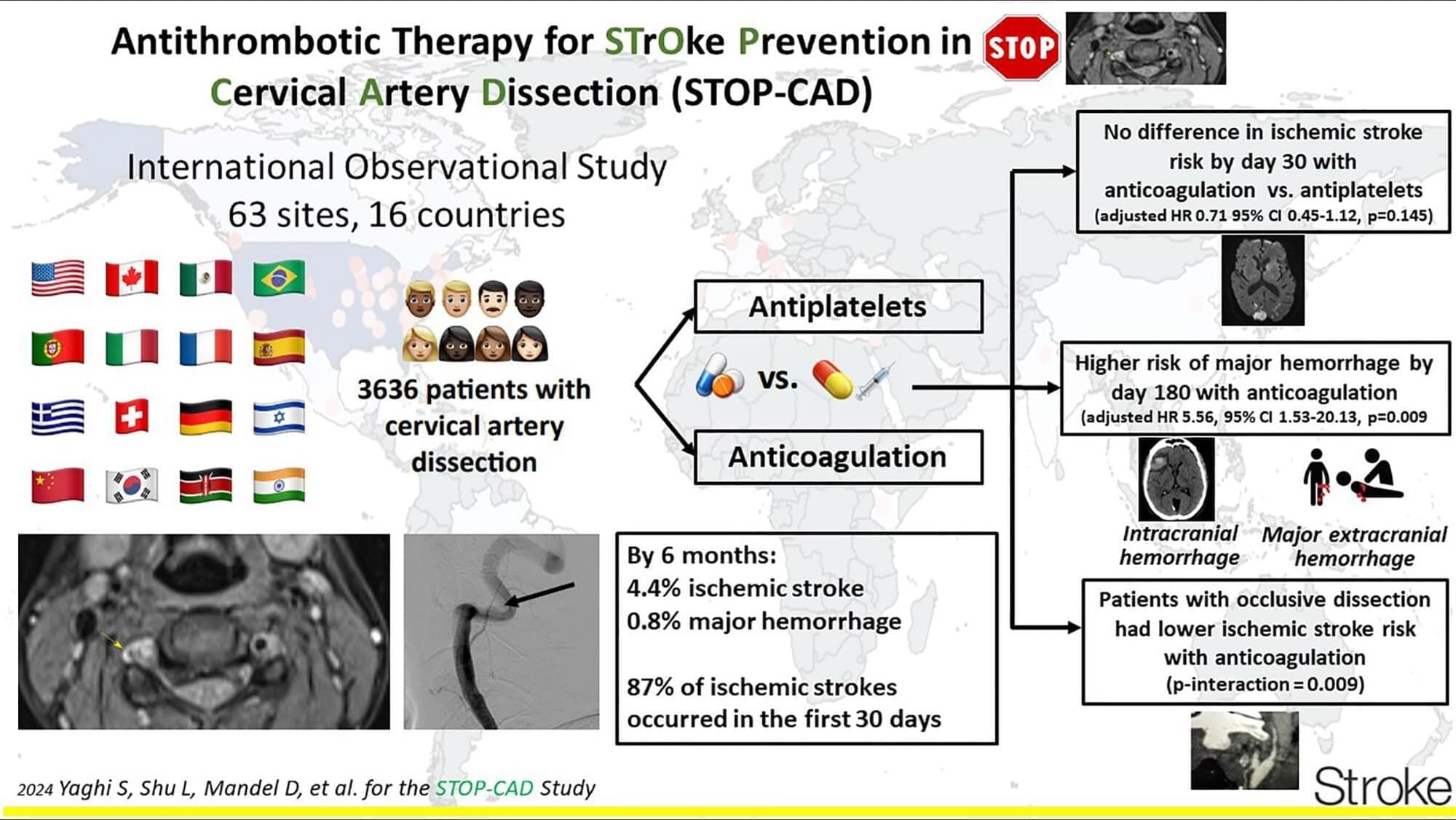
This was a multicenter international retrospective observational study (63 sites from 16 countries; Figure S1) that included patients presenting to an acute care hospital and diagnosed with CAD without concomitant major trauma. We identified adult patients aged ≥18 years with CAD based on International Classification of Diseases, Ninth Revision codes (443.21 and 443.24),8,9 International Classification of Diseases, Tenth Revision codes (I77.71, I77.74, and I77.75),10 or from institutional registries. These codes have been used or validated in prior studies.8–10
The patients’ vascular neuroimaging studies were reviewed by site principal investigators, and only those with clinical suspicion for CAD and imaging confirmation were included. Imaging confirmation required the presence of at least one of the following imaging features: crescent-shaped hyperintensity in the vessel wall indicating an intramural hematoma; a double lumen sign; the presence of a dissecting pseudoaneurysm, intimal flap, or vessel irregularity; or flame-shaped or tapering stenosis or occlusion of the artery at a typical dissection site and without evidence of atherosclerotic changes. Imaging reports, when available, were reviewed by neurologists at the lead site to confirm a dissection diagnosis.
We excluded patients with incidental chronic dissection, those with major head or neck trauma within the previous 4 weeks (eg, causing skull or cervical fractures or hemorrhage), those with a dissecting aneurysm causing primary subarachnoid hemorrhage, and those with iatrogenic dissection.
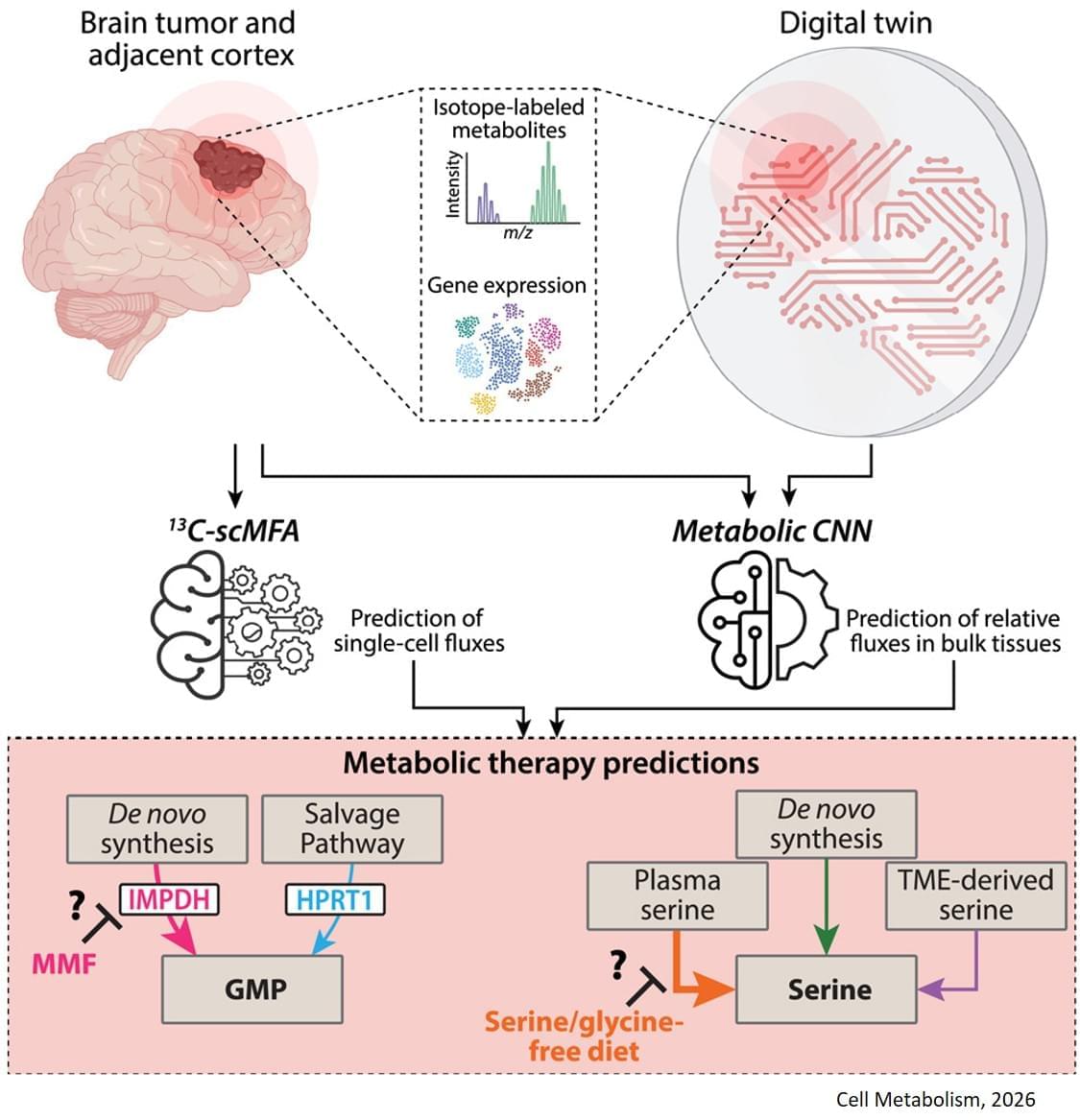
The study, published in Cell Metabolism, builds on previous research showing that some gliomas can be slowed down through the patient’s diet. If a patient isn’t consuming certain protein building blocks, called amino acids, then some tumors are unable to grow. However, other tumors can produce these amino acids for themselves, and can continue growing anyway. Until now, there was no easy way to tell which patients would benefit from dietary restrictions.
The digital twin’s ability to map metabolic activity in tumors also helped determine whether a drug that prevents tumors from producing a building block for replicating and repairing DNA would work, as some cells can obtain that molecule from their environments.
To overcome challenges in mapping tumor metabolism inside the brain, the team developed a computer-based “digital twin” that can predict how an individual patient’s brain tumor will react to each treatment.
“Typically, metabolic measurements during surgeries to remove tumors can’t provide a clear picture of tumor metabolism—surgeons can’t observe how metabolism varies with time, and labs are limited to studying tissues after surgery. By integrating limited patient data into a model based on fundamental biology, chemistry and physics, we overcame these obstacles,” said a co-corresponding author of the study.
The digital twin uses patient data obtained through blood draws, metabolic measurements of the tumor tissue and the tumor’s genetic profile. The digital twin then calculates the speed at which the cancer cells consume and process nutrients, known as metabolic flux.
“This is the first time a machine learning and AI-based approach has been used to measure metabolic flux directly in patient tumors,” said a co-first author of the study.
The researchers built a type of deep learning model called a convolutional neural network and trained it on synthetic patient data, generated based on known biology and chemistry and constrained by measurements from eight patients with glioma who were infused with labeled glucose during surgery. By comparing their computer models with different data from six of those patients, they found the digital twins could predict metabolic activity with high accuracy. In experiments conducted on mice, the team confirmed that the diet only slowed tumor growth in mice that the digital twin had identified as good candidates for the treatment.
The nurse dimmed the lights and checked the drip, a quiet beep keeping time in the corner of the oncology ward. On the bed, a young man in a faded band T‑shirt scrolled his phone, pretending not to notice his mother’s eyes darting between the monitor and the doctor at the door. Cancer, once again, was a ghost in the room – everywhere and nowhere, invisible yet controlling every breath.
The doctor took a breath of his own before speaking. This time, he said, they had something different. Not a bigger hammer. A smarter trick.
A way to make the ghost show its face.
These mini brain organoids resemble the developing cortex — the area of our brains that think, feel and store memories.
This advancement will help us learn more about conditions like dementia, but what if we go too far and they become conscious?
Hear the full story on The world, the universe and us, a news podcast for the insatiably curious, hosted by Rowan Hooper and Penny Sarchet.
Tap link in bio to learn more
https://www.newscientist.com/podcasts/
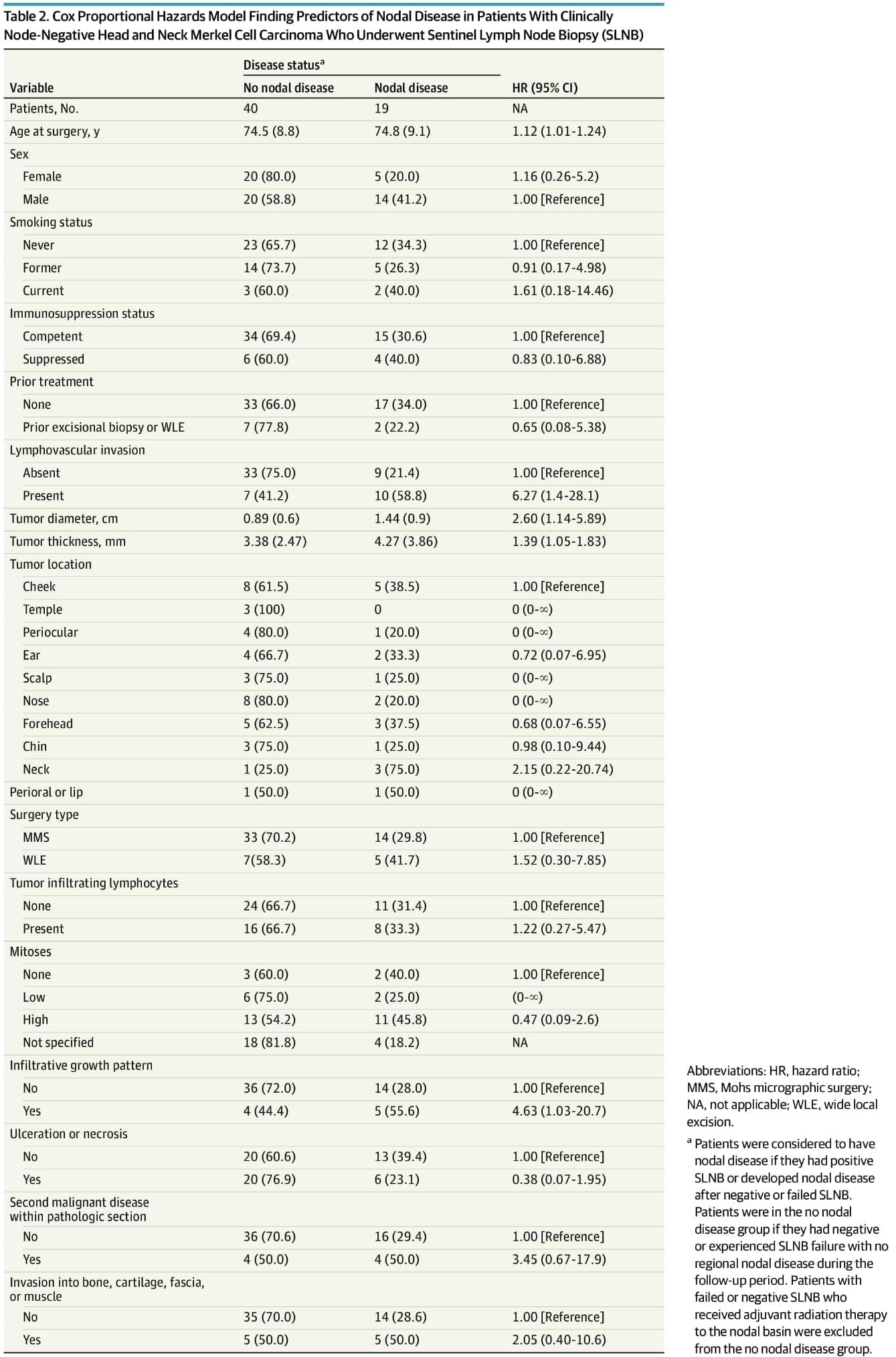
Between 34% and 44% of MCC cases arise in the head and neck region.1,3-6 This location presents unique challenges for SLNB due to complex lymphatic drainage patterns and proximity to critical anatomical structures.7,8 Accurate nodal staging is especially important because head and neck MCCs (HN MCC) may have worse prognoses than non−HN MCC.9,10
Prior studies of SLNB and HN MCC have been limited by small sample sizes (often 10 patients) and have reported relatively high false-negative rates.8, 11-13 A multisite retrospective study14 characterizing predictors of FN SLNB in stage I/II MCC included 214 patients with HN MCC and found a 39% FN rate in the HN cohort but did not otherwise analyze this cohort separately or describe the unique considerations associated with this region. High SLNB failure rates have also been observed in HN melanoma and squamous cell carcinoma and been largely attributed to anatomic complexity.15-17
In this large cohort study of patients with clinically node-negative HN MCC who underwent SLNB, we sought to evaluate SLNB accuracy within this anatomically complex region and to identify factors associated with SLNB failure and nodal disease. Our goal was to inform staging practices and refine risk stratification strategies to improve treatment planning and outcomes for this high-risk population.
(Cell Reports 16, 2641–2650; September 6, 2016)
The original version of this article, published on August 25, 2016, included an incorrect immunoblot image. The anti-H3 loading control shown in Figure 2C was inadvertently duplicated from a separate project during manuscript preparation. This was an unintentional oversight.
Normally, the authors would replace the incorrect image with the correct one. However, because the experiments were performed more than 10 years ago, the original image/film could not be located. This is consistent with the authors’ institutional data-retention policy, which requires data to be kept for only three years after a project’s completion. After discussion, the authors and editors agreed that removing the immunoblot images from Figure 2C would prevent confusion for future readers without changing the article’s central conclusions.
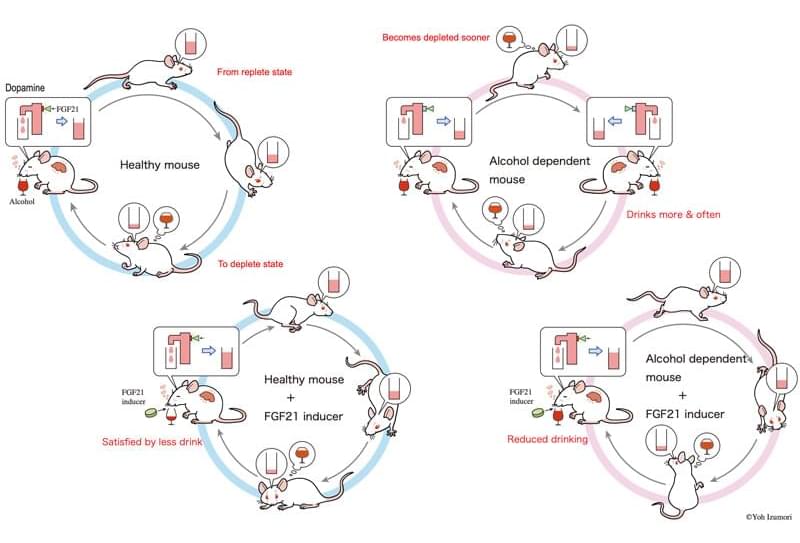
While investigating the FGF21-oxytocin-dopamine system, a mechanism that regulates sugar appetite, a team of researchers at Kyoto University noticed reports suggesting that the protein FGF21 may regulate alcohol ingestion.
The team’s original aim had been to address sugar appetite in lifestyle-related diseases, but since alcohol is a fermented product of sugar, they speculated that perhaps the body contains a system that recognizes both alcohol and sugar as the same entity.
Alcohol consumption and intervention challenges Excessive alcohol consumption is a major global health issue, and effective countermeasures for prevention and treatment are limited. Patients with alcohol dependence generally have a low adherence to pharmaceuticals, and many avoid drug treatment because it deprives them of the pleasure of drinking.
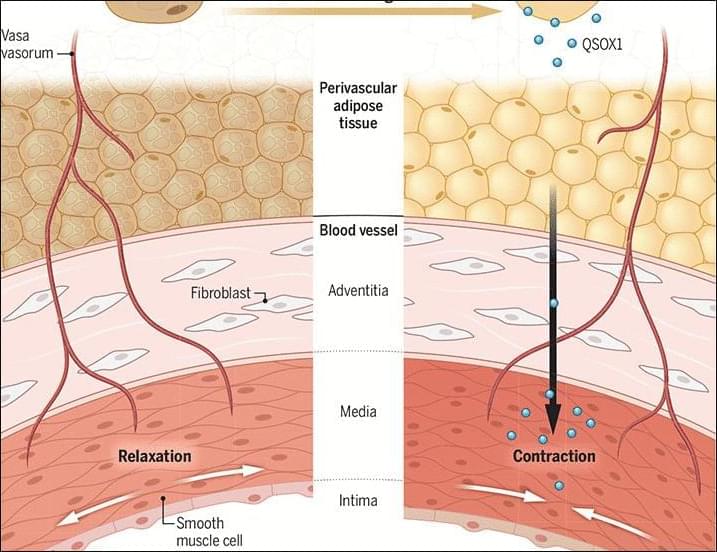
Beige fat surrounding blood vessels actively works to keep high blood pressure in check, according to a new Science study in mice, promoting healthy vascular function even during obesity.
The findings support the notion that therapeutic activation of thermogenic fat tissue could help reduce the risk of cardiovascular disease.
Learn more in this Science vPerspective.
A protein in perivascular fat cells protects mice against hypertension.
Mandy O. J. Grootaert and Aernout Luttun Authors Info & Affiliations
Science
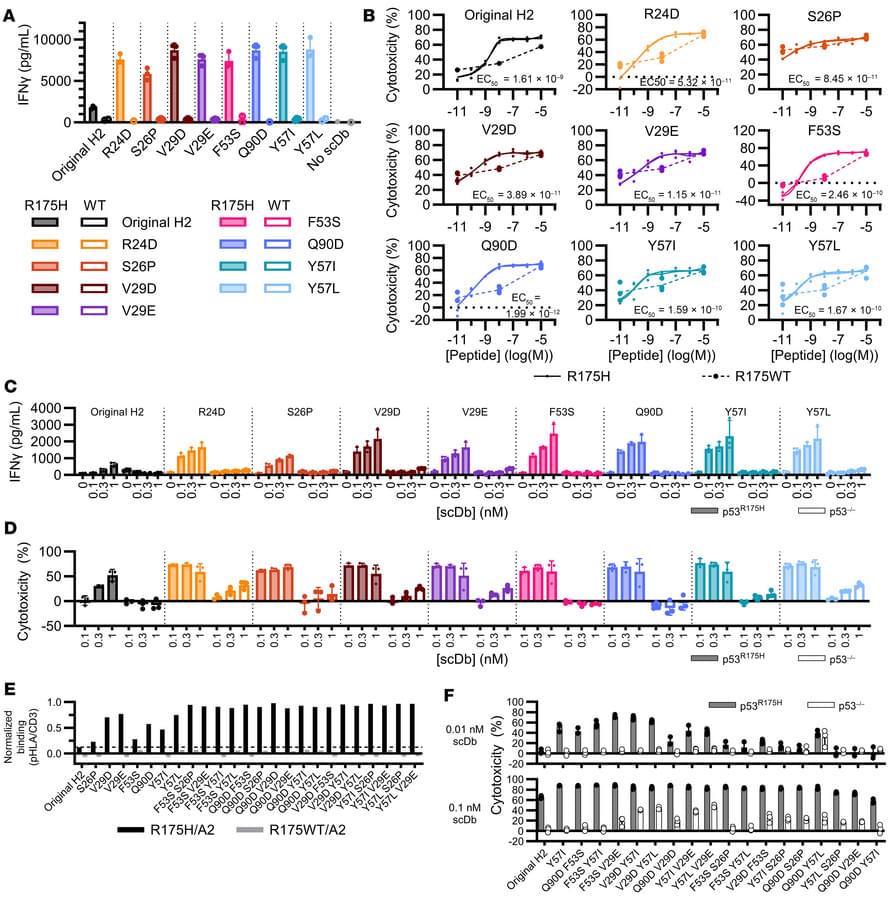
Here, Bert Vogelstein & team report that increasing binding affinity to a p53 R175H neoantigen improves anti-cancer activity for T cell-engaging bispecific antibodies and conversely decreases function for CAR T cells.
1Ludwig Center, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University School of Medicine, Baltimore, Maryland, USA.
2Howard Hughes Medical Institute, Chevy Chase, Maryland, USA.
3Lustgarten Pancreatic Cancer Research Laboratory and.
4Bloomberg~Kimmel Institute for Cancer Immunotherapy, Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University School of Medicine, Baltimore, Maryland, USA.