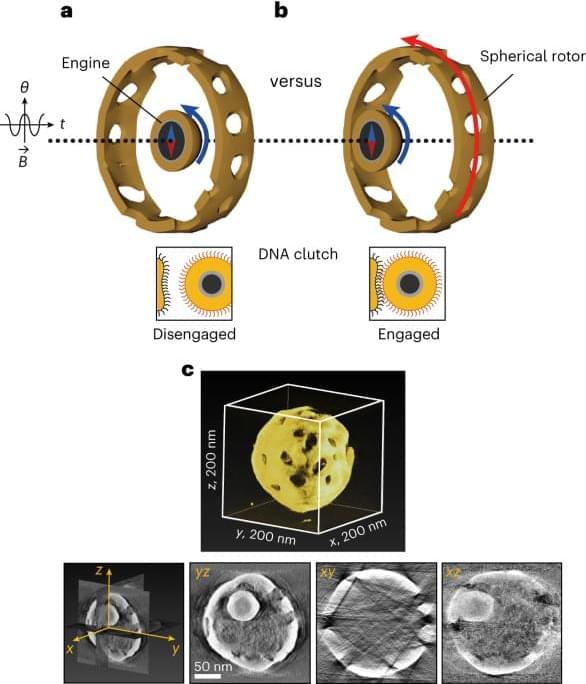
The nanospace confinement of a magnetic nanoparticle within a porous cage, coupled with an encodable DNA clutch interface, enables a remotely powered and controlled rotary nanomotor that is autoresponsive to its microenvironment.

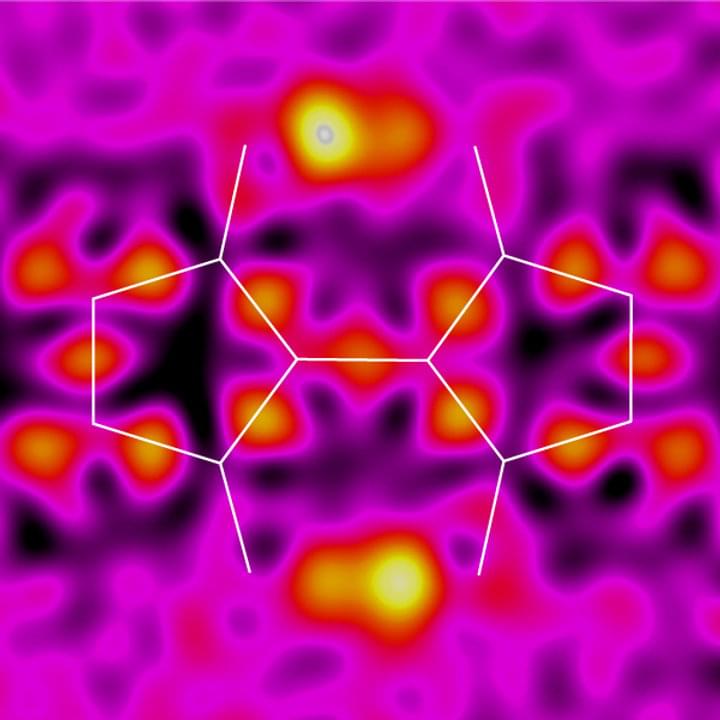
Chemists at RIKEN have developed a method for making synthetic derivatives of the natural dye indigo that doesn’t require harsh conditions. This discovery could inspire advances in electronic devices, including light-responsive gadgets and stretchy biomedical sensors.
Semiconductors based on organic molecules are attracting much interest because—unlike conventional rigid semiconductors based on silicon—they could be flexible, ductile and lightweight, opening up new possibilities for designing semiconductor devices.
Organic molecules also have the advantage of realizing a broad range of structures. “Organic semiconductors have flexibility in molecular design, enabling them to adopt new functionalities,” says Keisuke Tajima of the RIKEN Center for Emergent Matter Science, who led the research.

A new fusion of materials, each with special electrical properties, has all the components required for a unique type of superconductivity that could provide the basis for more robust quantum computing. The new combination of materials, created by a team led by researchers at Penn State, could also provide a platform to explore physical behaviors similar to those of mysterious, theoretical particles known as chiral Majoranas, which could be another promising component for quantum computing.
The new study appears in the journal Science. The work describes how the researchers combined the two magnetic materials in what they called a critical step toward realizing the emergent interfacial superconductivity, which they are currently working toward.
Superconductors—materials with no electrical resistance —are widely used in digital circuits, the powerful magnets in magnetic resonance imaging (MRI) and particle accelerators, and other technology where maximizing the flow of electricity is crucial.
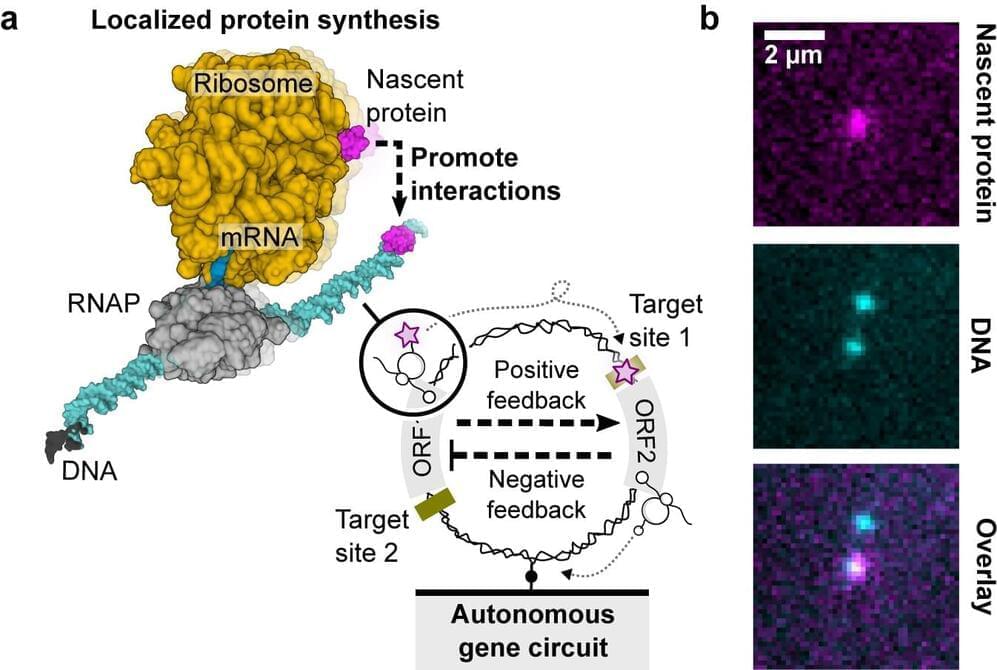
In a new Nature Communications study, researchers have explored the construction of genetic circuits on single DNA molecules, demonstrating localized protein synthesis as a guiding principle for dissipative nanodevices, offering insights into artificial cell design and nanobiotechnology applications.
The term “genetic circuit” is a metaphorical description of the complex network of genetic elements (such as genes, promoters, and regulatory proteins) within a cell that interact to control gene expression and cellular functions.
In the realm of artificial cell design, scientists aim to replicate and engineer these genetic circuits to create functional, self-contained units. These circuits act as the molecular machinery responsible for orchestrating cellular processes by precisely regulating the production of proteins and other molecules.

— Anti-aging vaccine shows promise in mice — will it work in humans?
“This research is still in the early stages and may be a number of years from being available to patients,” Dr. David Pinato, a clinician scientist at Imperial College London’s Department of Surgery & Cancer and a consultant medical oncologist at Imperial College Healthcare NHS Trust, said in the statement. “But this trial is laying crucial groundwork that is moving us closer towards new therapies that are potentially less toxic and more precise.”
The first person treated in the U.K. arm of the trial wishes to remain anonymous, but said “I was pleased to be offered a chance to take part in a new trial.”

Could cannabis use lead to anxiety disorders? This is what a recent study published in EClinicalMedicine hopes to address as a team of researchers led by the Ottawa Hospital Research Institute examine the connection between cannabis use and anxiety disorders by patients who have had emergency department visits. While this study was limited to Canada, it holds the potential to help researchers, legislators, and the public better understand the long-term risks of cannabis on mental health disorders throughout the world.
For the study, the researchers analyzed health records from 2008 to 2019 for 12,099,144 individuals between 10 and 105 years of age across Ontario, Canada who had no prior history of being diagnosed with an anxiety disorder from any hospital visit and compared this to the general population. In the end, the researchers found that 27.5 percent of individuals who visited a hospital for cannabis use were diagnosed with an anxiety disorder within three years of their hospital visit, whereas only 5.6 percent of the general population experienced the same. Additionally, they found that individuals who visited a hospital for cannabis use also visited a hospital for an anxiety disorder within three years of the initial hospital visit, whereas only 1.2 percent of the general population experienced the same.
“Our results suggest that individuals requiring emergency department treatment for cannabis use were both at substantially increased risk of developing a new anxiety disorder and experiencing worsening symptoms for already existing anxiety disorders,” said Dr. Daniel Myran, who is a Faculty of Medicine in the Department of Family Medicine at the University of Ottawa and lead author of the study.
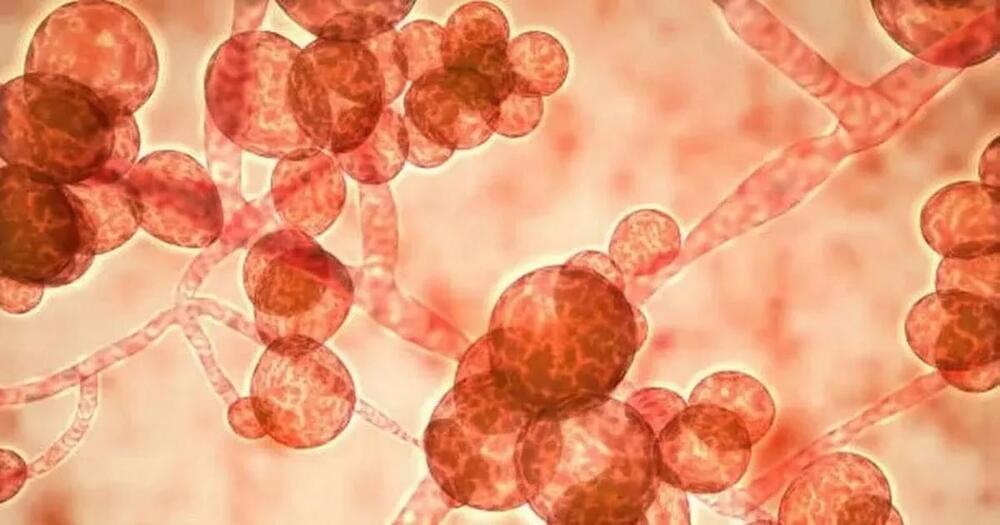
C. auris, a type of yeast that can cause severe illness, is a “global health threat”, according to experts.
Bookmark


Massachusetts startup Elemind has raised $12 million to read brainwaves and treat people for sleep disorders, long-term pain, tremors, and to speed up learning rates. Clinical trials show the company’s wearable device can accelerate sleep up to 70% faster, reduce tremors in patients with physiological shaking up to 50%, and boost learning rates.
“We use a wearable neurotech device to read the brain in real time and intercept it in real time with something called neurostimulation,” Elemind co-founder and CEO Meredith Perry told me on a recent TechFirst podcast. “That’s using sound or light or vibration or electricity to stimulate the brain. And when we do that, we can actually guide the brain precisely, and that leads to a behavior change. So like a drug, but much smarter and without the side effects.”