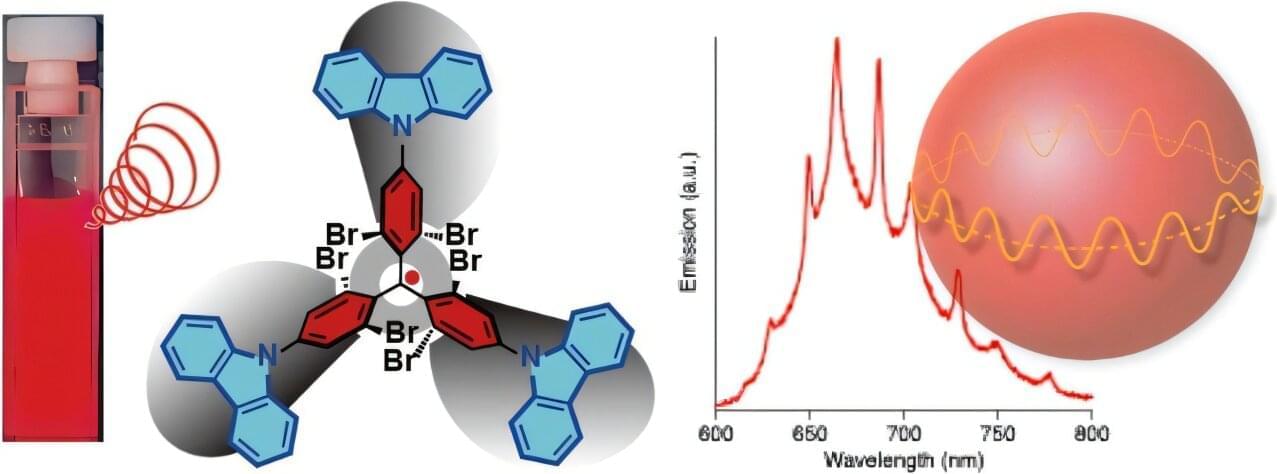
Circularly polarized light has properties that make it useful in a growing range of technologies, from next-generation 3D displays to bioimaging tools that can detect signals deep within living tissues. One way to produce this kind of light is with the help of chiral molecules—compounds that have a mirror-image form to which they cannot be perfectly superimposed. Among these, small organic molecules (SOMs) offer tunable emission wavelengths.
Luminescent radicals represent a promising type of SOM for red and near-infrared circularly polarized luminescence (CPL) emission. One particular family of radicals, tris(2,4,6‑trichlorophenyl)methyl (TTM)‑based radicals, is inherently chiral and a natural candidate for CPL.
In practice, however, these molecules fall short on multiple fronts, with tradeoffs between stable chirality, high emission efficiency, and durability under operating conditions.