Telomere structure shapes cell fate decisions.
Telomere loops (t-loops) are dynamic DNA structures, remodelled during the cell cycle and stress, rather than static protective caps.
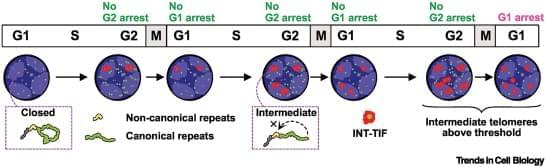
The three-state model defines closed, intermediate, and uncapped telomeres, linking intermediate telomeres to programmed, fusion-resistant deprotection, which activates checkpoints without genome instability.
Mitotic arrest-dependent telomere deprotection is an active pathway in which Aurora B kinase drives t-loop unwinding without telomere shortening.
Aurora B kinase phosphorylation reprograms shelterin components (TRF1 and TRF2), enabling BTR-mediated t-loop dissolution and paradoxically converting protective factors into facilitators of deprotection.
T-loop dynamics reframe telomeres as responsive signalling hubs that couple chromosome architecture to genome surveillance and cell fate control. sciencenewshighlights ScienceMission https://sciencemission.com/T-loop-dynamics
Telomeres are nucleoprotein elements bound by shelterin that protect chromosome ends from DNA damage signalling and inappropriate repair. A defining architectural feature is the telomere loop (t-loop), a lariat structure formed by 3’ overhang invasion into duplex telomeric DNA, which sequesters chromosome ends from damage recognition. T-loop stability is disrupted by the loss of the shelterin component TRF2, and progressive telomere shortening during ageing is predicted to compromise t-loop maintenance. In addition to intrinsic erosion, an active, shelterin-directed mechanism unwinds t-loops during mitotic arrest. This mitotic arrest-dependent telomere deprotection promotes mitotic death, requires Aurora B kinase-dependent shelterin phosphorylation and the BTR complex, and is opposed by WRN.