Iron and oxygen bind together throughout the body. Most famously, iron binds dioxygen, or two oxygens paired with each other, in hemoglobin that transports oxygen through blood. But iron-oxo compounds, as they’re called, are found in many other places throughout the body. For example, the highly reactive iron-oxo is used in liver enzymes that metabolize drugs.
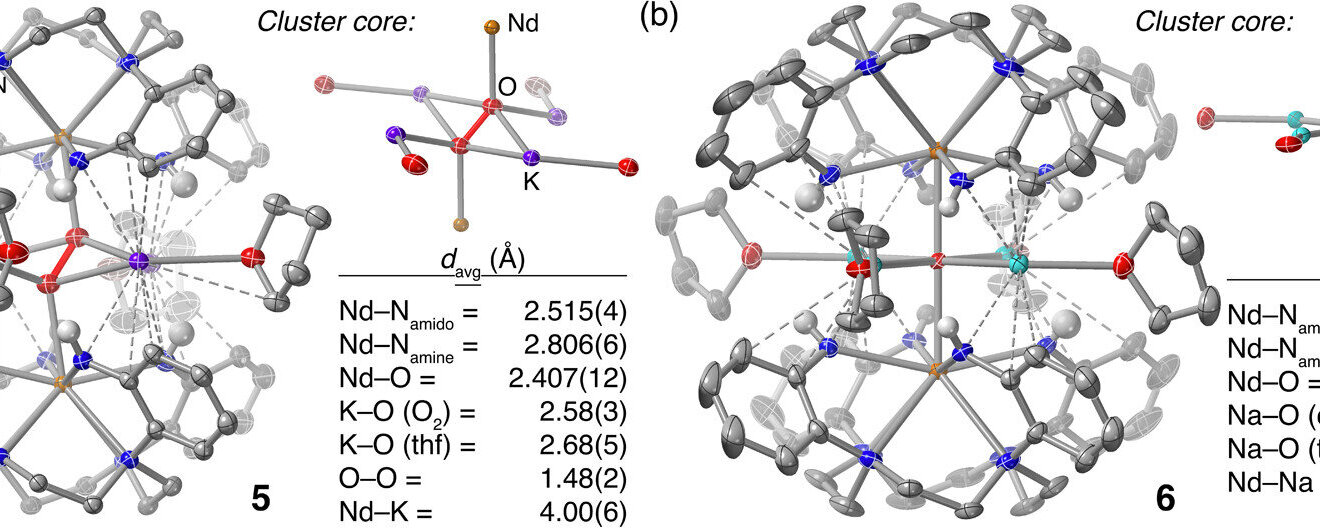
Rice University chemist Raúl Hernández Sánchez was interested in how oxygen could react with other types of metals—ones that reside on the lowest section of the periodic table, known as f-block metals, with lanthanides on the upper row and actinides on the lower.
If lanthanides could bind with oxygen, he theorized, it would form a highly reactive lanthanide-oxo compound that potentially could be used as a synthetic replacement for iron-oxo, opening up a new toolbox for small molecule chemists interested in studying these biological reactions.