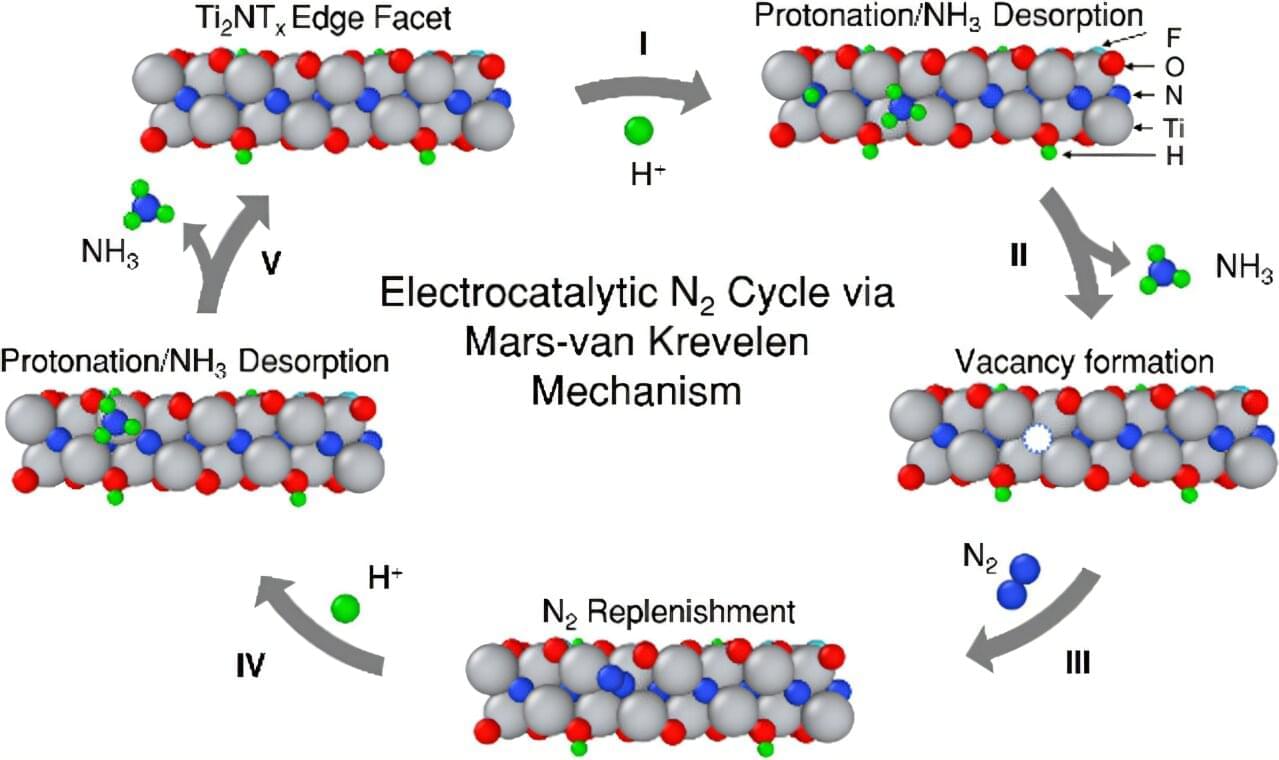
With a new electrochemical synthesis via an electrochemical nitrogen reduction reaction (NRR), achieving carbon-free ammonia production is closer to reality through work from Drs. Abdoulaye Djire and Perla Balbuena, chemical engineering professors at Texas A&M University, and graduate students David Kumar and Hao En Lai. A topic outlined in their recent paper published in the Journal of the American Chemical Society introduces NRR, which produces ammonia in a cleaner and simpler way by using renewable electricity.
The research branches off of the team’s previous work, where they looked further into enabling two-dimensional materials in renewable energy.
“The current process of making ammonia is energy intensive and emits a lot of carbon dioxide, so if you can make ammonia electrochemically, then you can avoid these two negative effects,” Djire said. “During the electrochemical NRR process, water provides the hydrogen atoms, which combine with nitrogen from the air to form ammonia, all powered by electricity.”