Using a renewable energy source has multiple benefits, including reducing harmful emissions and dependence on fossil fuels while increasing efficiency. But many renewable energy sources have a higher cost than fossil fuels due to the materials needed to make them usable, such as platinum group metals (PGMs), and the high cost of storage.
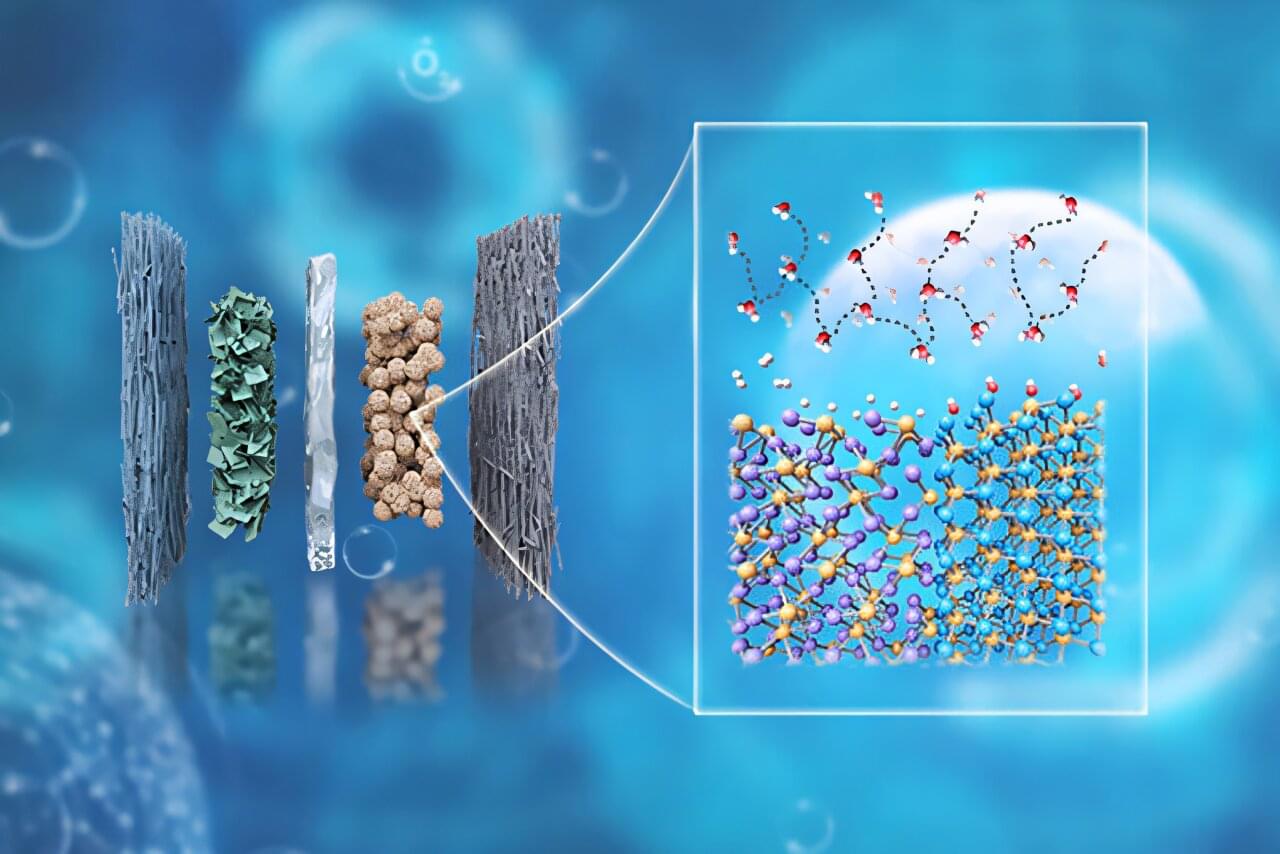
A team of researchers led by Gang Wu, a professor of energy, environmental and chemical engineering at the McKelvey School of Engineering at Washington University in St. Louis is working to change that. The team is creating a heterostructure catalyst for an anion-exchange membrane water electrolyzer (AEMWE) that splits water into hydrogen and oxygen using electricity from renewable sources. They created the catalyst with two phosphides that gave them an efficient method to extract hydrogen, a valuable yet low-cost source of zero-emissions fuel. The study is published in the Journal of the American Chemical Society.
Wu’s team has been looking for alternatives to catalysts that use expensive platinum group metals. In this research, their idea began with using sunlight, wind or water to create electricity that they could then use to separate hydrogen from water.
Why visitors still use to read news papers when in this technological globe everything is existing on web?
https://share.google/Vl6QkTAjlLthvLwwn