In adults with methamphetamine use disorder, mirtazapine reduced methamphetamine use by approximately two more days per month vs placebo, with no unexpected safety concerns.
Main Outcomes and Measures The primary end point was the change in days of methamphetamine use in the past 28 days from baseline to week 12. Secondary end points were depression, insomnia, HIV risk behavior, quality of life, and methamphetamine-negative oral fluid samples.
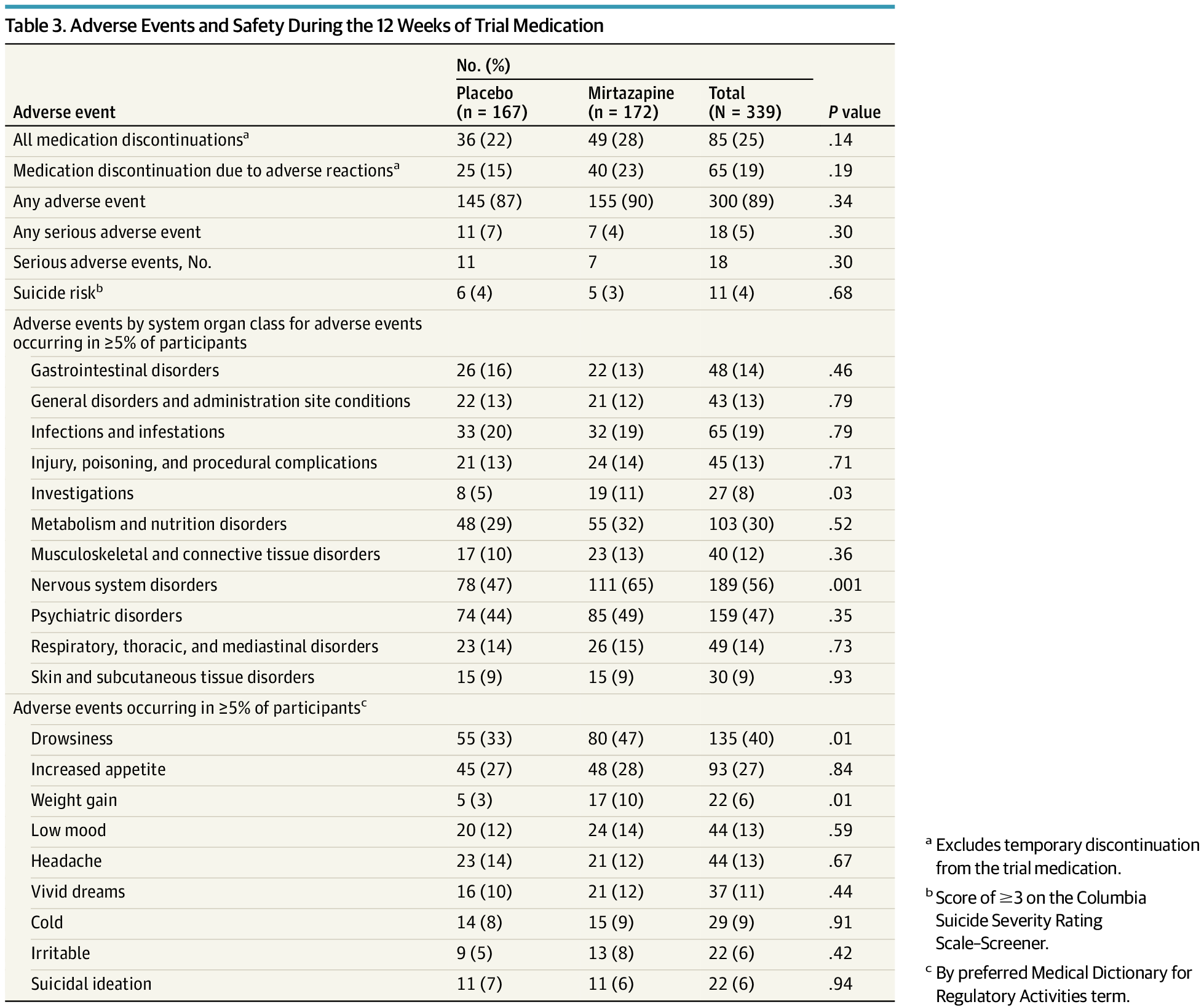
Results Of 344 participants randomized, 339 participants received the intervention (167 in the placebo group and 172 in the mirtazapine group). Mean (SD) age was 42.0 (8.6) years, 126 participants (37.2%) were female, and participants had used methamphetamine for a median (IQR) of 24 days (17−28) of the past 28 days at baseline. The mean reduction in days of methamphetamine use from baseline to week 12 was greater in the mirtazapine group (7.0 days of 28 days) than in the placebo group (4.8 days of 28 days; mean difference, 2.2 days; 95% CI, −4.2 to −0.2 days; P = .02). More participants in the mirtazapine group reported drowsiness (47% vs 33%) and weight gain (10% vs 3%). Forty participants (23%) discontinued mirtazapine due to adverse events compared to 25 participants (15%) in the placebo group. No significant effects of mirtazapine on secondary end points were found.
Conclusions and Relevance In this parallel-group randomized clinical trial, mirtazapine delivered in routine clinical practice reduced methamphetamine use in adults with methamphetamine use disorder. No unexpected safety concerns delivering mirtazapine in this setting were found; this finding has important clinical implications in the absence of any approved pharmacotherapies for methamphetamine use disorder.