New in JBC press|
asbmbJBC.
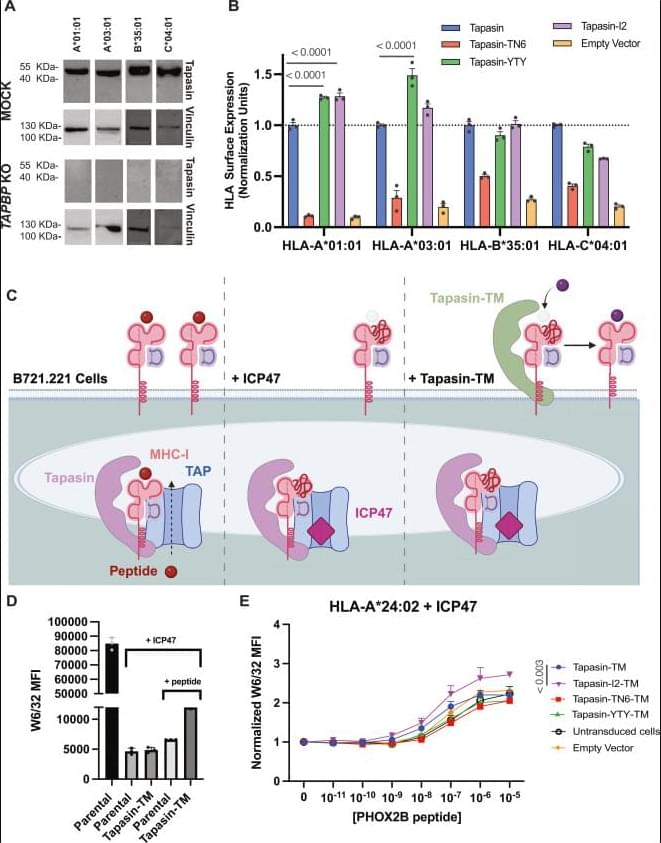
Human leukocyte antigen (HLA) proteins are extremely polymorphic, with different allotypes exhibiting a wide range of dependencies on the chaperone tapasin for peptide loading, expression, and stability at the cell surface. Given its central role in antigen processing, tapasin is frequently downregulated across viral infections and cancers, impairing antigen presentation and hindering the identification of therapeutically relevant peptide antigens. We hypothesized that elucidating the mutational tolerance of tapasin surfaces which mediate interactions with polymorphic HLA residues can provide a means for fine-tuning its chaperoning function and reveal mechanistic epitopes that underlie its function.