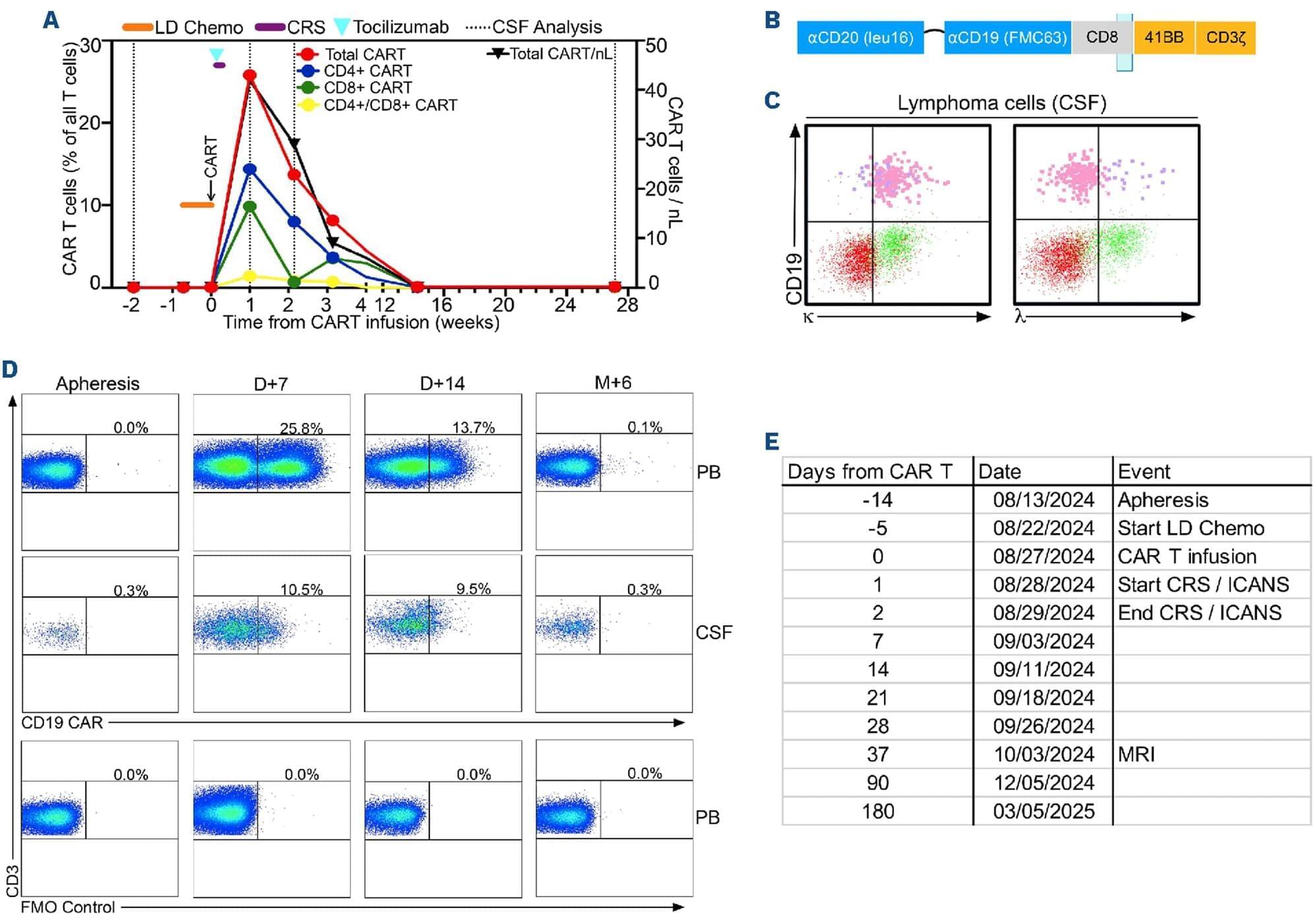
CAR T cells in primary CNS lymphoma.
Treatment options for primary central nervous system (CNS) lymphoma remain limited, particularly in relapsed or refractory disease. This case report explores the activity and CNS trafficking of tandem CD20/CD19 CAR T cells (zamtocabtagene autoleucel), addressing key questions about cellular therapy in CNS lymphoma and the potential role of dual-target CAR T strategies.
CD19-directed chimeric antigen receptor (CAR) T-cell (CAR T) therapy is a well-established treatment for B-cell malignancies, but accessibility, toxicities, lack of persistence, modest anti-tumor activity, restricted trafficking and tumor antigen escape are among its limitations.1 Concern for excessive neurotoxicity led to exclusion of patients with central nervous system (CNS) involvement from clinical trials, and primary CNS lymphoma (PCNSL) is an exclusion on the approved products’ labeling. PCNSL is a highly aggressive lymphoma, with a favorable response to initial chemotherapy/radiation, but compared with lymphomas outside the CNS, relapses are common, and survival is inferior. Moreover, there is a lack of approved standard care beyond first-line therapy and the prognosis for these PCNSL patients remains poor.2
In approximately one third of all B-cell lymphoma patients, resistance to CAR T and relapses are accompanied by CD19 downregulation. Preclinical evidence suggests that dual antigen-targeting may overcome this problem.3 To address antigen escape, the investigational anti-CD20/ anti-CD19 CAR T product MB-CART2019.1 (zamtocabtagene autoleucel [zamto-cel]) was designed. Zamto-cel is a dual-targeting tandem-CAR construct with scFv regions of anti-CD19 and anti-CD20 linked in sequence by a flexible interchain linker, followed by CD8, 4-1BB and CD3 ζ domains.4 Our group and others have evaluated zamto-cel in a pivotal phase II clinical trial (DALY II USA/ MB-CART2019.1; clinicaltrials gov. Identifier: NCT04792489) for the treatment of DLBCL patients who received at least two lines of treatment.5 In addition to addressing antigen escape/relapse zamto-cel is non-cryopreserved with a vein-to-vein time of 14 days, improving cell yield and potency.