This phase 2 nonrandomized clinical trial evaluated oral upadacitinib for severe immune checkpoint inhibitor–related dermatitis in patients with solid tumors.
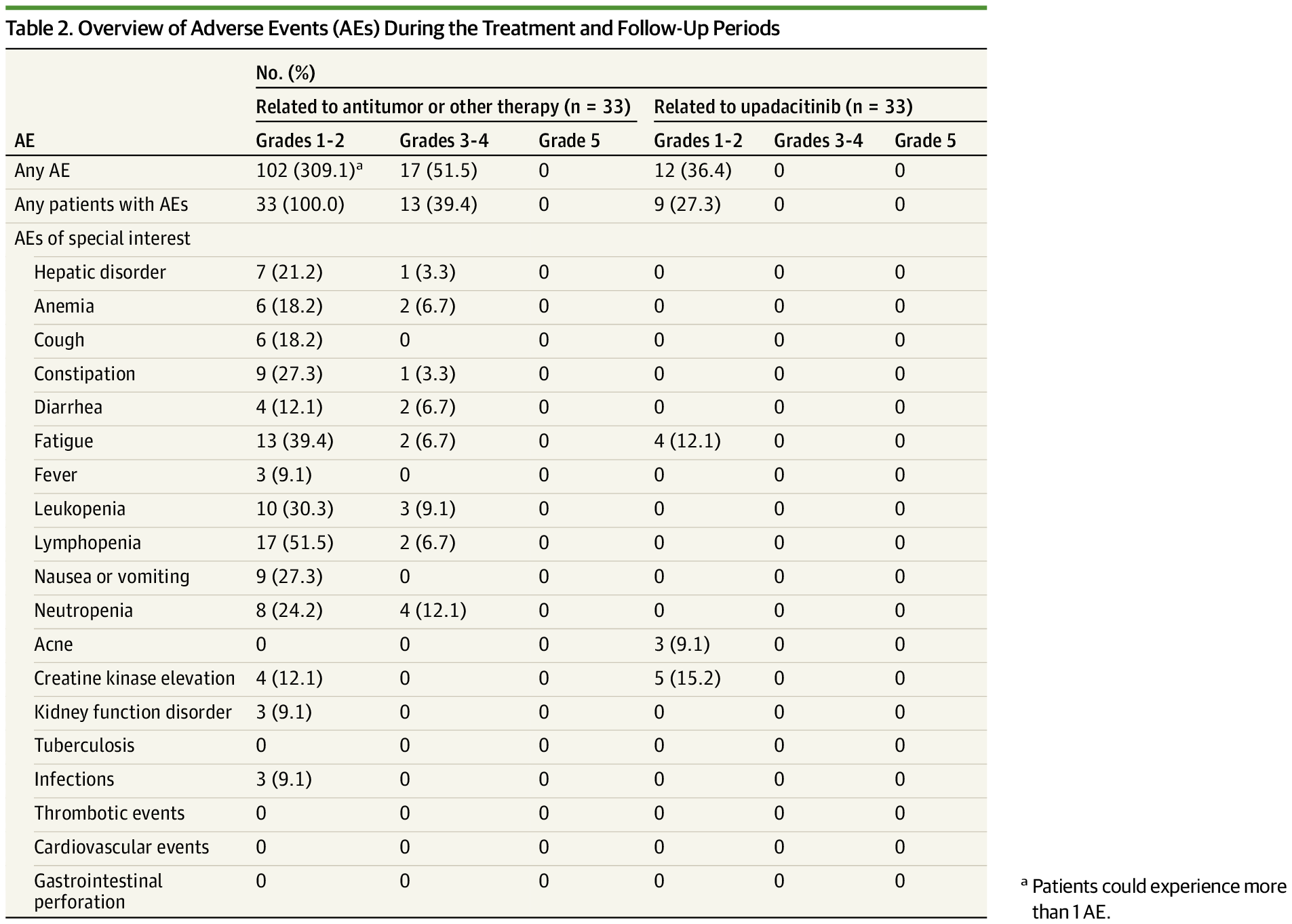
Rash resolution to grade ≤1 was achieved in all participants within 28 days, and pruritus improvement was reported within 1 day of initiation. Upadacitinib was well tolerated, with no serious treatment-related adverse events.
The majority of patients were able to continue immune checkpoint inhibitor therapy as scheduled, indicating the potential of upadacitinib as a safe and effective option for managing severe Dermatitis induced by immunotherapy.
This nonrandomized clinical trial examines the safety and efficacy of oral upadacitinib in the treatment of patients with severe immune checkpoint inhibitor–related dermatitis.