Ultrastructural Preservation of a Whole Large Mammal Brain (bioRxiv, 2026) ⚠️ Preprint – not yet peer-reviewed.
A 2026 preprint builds on over a decade of brain preservation research, demonstrating that whole mammalian brains (pigs) can be preserved with remarkable structural fidelity under near–real-world, end-of-life conditions.
The study refines aldehyde-stabilized cryopreservation (ASC)—a technique previously recognized by the Brain Preservation Foundation. This method combines chemical fixation (aldehydes), cryoprotectants, and controlled cooling to prevent ice damage and preserve neural structure at the nanoscale. — What the study shows.
Whole pig brains preserved with intact cellular and synaptic architecture.
Preservation remains viable even with delayed postmortem intervals (~10 minutes)
Tissue remains perfusable and structurally stable after fixation.
Protocol moves toward clinically realistic implementation, not just lab conditions.
👉 In short: This is less about proving preservation is possible—and more about making it practical in real-world scenarios. –
🧠 Chief Scientist Commentary (Nectome)
Nectome’s Chief Scientist, Borys Wróbel, expands on the implications:
Claims preservation quality is likely sufficient to retain memory and psychological information.
Suggests future pathways like whole brain emulation (WBE) or even physical reconstruction.
Notes the method can support long-term storage (liquid or vitrified states)
Emphasizes ethical framing: consent-based preservation as a “bridge” to future technologies.
He also clarifies technical points:
Aldehydes create crosslinks between proteins (not cells)
Preservation can begin minutes after death (~10 min demonstrated)
Long-term storage may occur above or far below freezing depending on method –
⚠️ Critical distinction: Evidence vs interpretation.
✅ Supported by the preprint.
High-quality structural preservation of brain tissue.
Feasibility in large mammals under realistic timing constraints.
⚠️ Not demonstrated (still hypothetical)
That preserved structure = recoverable memory or identity.
That future technology can reconstruct a mind.
Any form of revival or consciousness restoration.
The key assumption remains unproven:
That preserving synaptic structure is sufficient to preserve the “self” –
This work represents an important shift:
From: “Can we preserve the brain?” To: “Can we preserve it in a way that could actually be used in humans?”
It advances:
Clinical feasibility.
Whole-brain / whole-body preservation workflows.
Long-term biostasis concepts.
But it also sits at the boundary between:
Established neuroscience (structure preservation)
Speculative future tech (mind reconstruction / digital continuity) –
Final takeaway.
A real technical milestone in brain preservation—but claims about preserving “you” remain a hypothesis, not a demonstrated outcome.
Preprint.
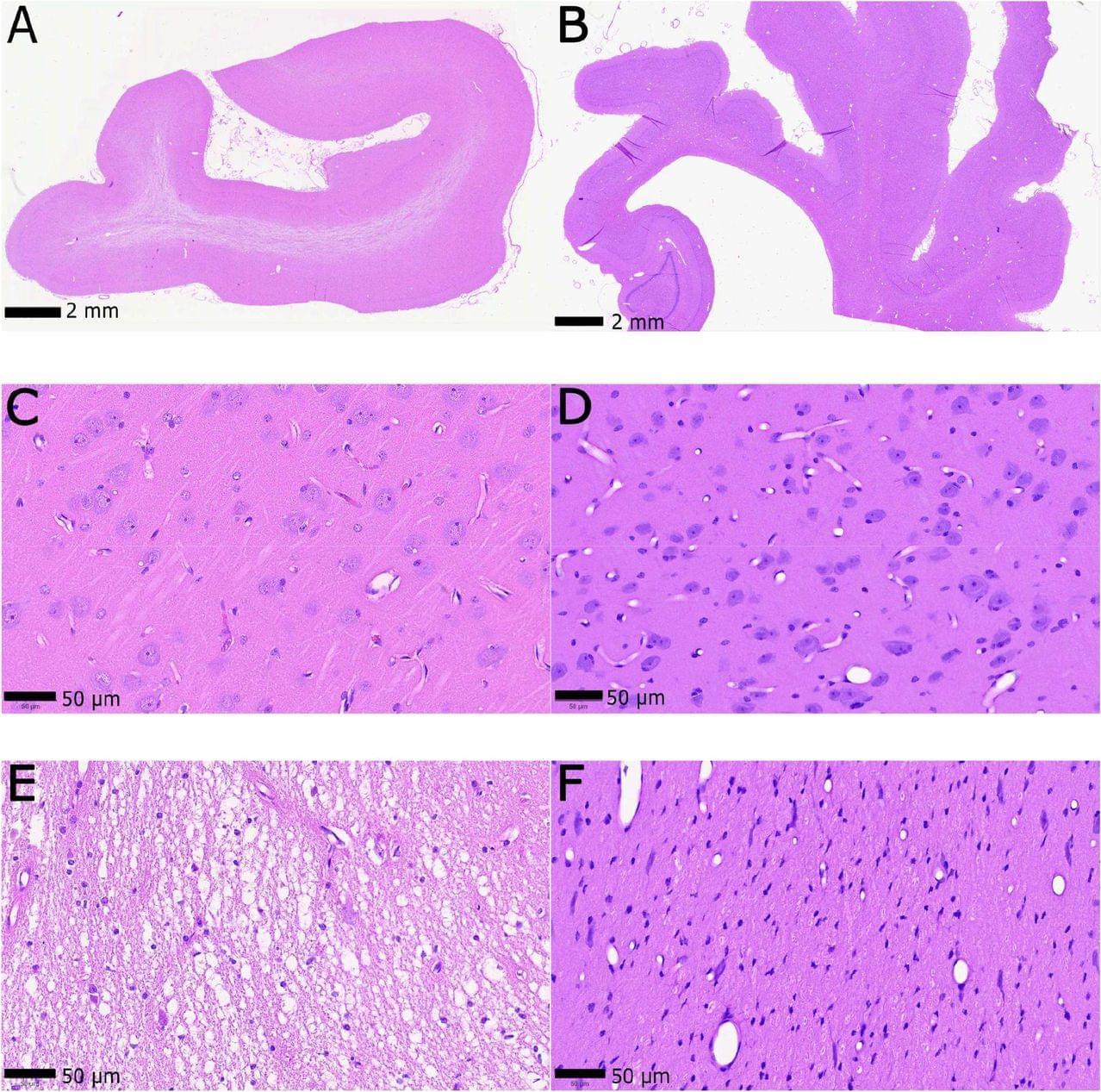
Building a high-fidelity computational model of the whole human brain will require preservation of the ultrastructure at the level of the entire organ, post-mortem. For such a model to reflect as closely as possible the brain in the living state, artifacts that arise during both the agonal phase and the postmortem interval will need to be minimized. This is potentially feasible if a terminally-ill patient donates their brain for research following physician-assisted death. In this paper, we modify a protocol for aldehyde-stabilized cryopreservation to make it compatible with physician-assisted death. We use pigs as a model, which resemble humans in cardiovascular and brain anatomy. Aldehyde-stabilized cryopreservation was designed to provide superior structural preservation of brains of any size, across all anatomical scales, compatible with diverse analytical assays and long-term storage without ultrastructural degradation. We demonstrate, with light microscopy and volume electron microscopy, that our brain preservation protocol results in connectomically traceable whole brains and propose an economically feasible storage modality that is expected to maintain stability of ultrastructure and macromolecules in the brain even for thousands of years. Most importantly, we establish that 14 min is the approximate length of the perfusability window—the time after the cardiac arrest during which blood washout needs to be initiated so that the brain ultrastructure is preserved.
The human brain is hierarchically organized across spatial scales from nanometers (macromolecules, their complexes), through micrometers (synapses, cells), and millimeters (neuronal circuits), to decimeters (whole-brain networks) [1]. It contains an enormous number of cells and synapses—estimates suggest an average of 60–100 billion neurons (and a similar number of glial cells), with the total number of synapses in hundreds of trillions [2,3]. Understanding how the brain generates internal representations, controls movement, stores and retrieves memories, and supports cognition and consciousness will require bridging all these spatial scales, as these properties emerge at the level of the whole organ and, arguably, above.