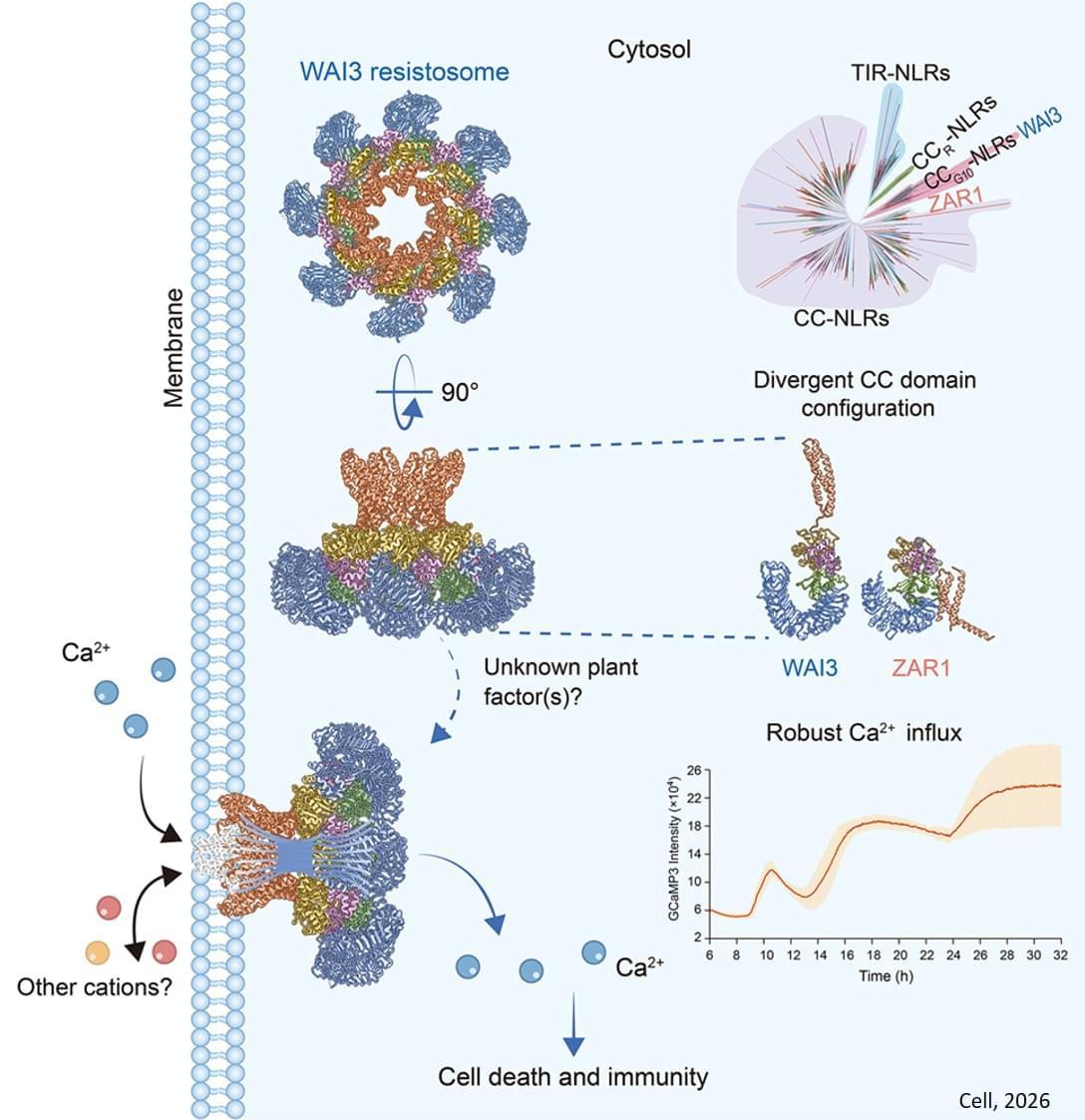
After activation, these NLRs form multi-protein complexes—called resistosomes—that carry out the immune response. Studies have shown that certain resistosomes are pentameric (e.g., ZAR1 and Sr35), whereas others are hexameric (e.g., NRC2 and NRC4). These complexes initiate immune responses by triggering calcium (Ca2+) influx into the cytoplasm. However, the G10 type of CC-NLR (CCG10-NLR) immune receptors constitutes a unique clade among CC-NLRs and its activation mechanism has remained poorly understood.
Now, in a study published in Cell, a research team has revealed a novel octameric resistosome formed by an activated wheat CCG10-NLR immune receptor, which induces Ca2+ influx and immune responses through a unique channel architecture.
The researchers identified the Wheat Autoimmunity 3 (WAI3) gene, which encodes a CCG10-NLR protein. Subsequent analysis revealed that a gain-of-function (GOF) single amino acid mutation in the leucine-rich repeat (LRR) domain leads to autoactivation, providing an opportunity to study the activation mechanism of CCG10-NLR.
After expressing the WAI3 proteins in Nicotiana benthamiana, the researchers used cryo-electron microscopy to resolve the octameric structure of the activated WAI3 resistosome—marking the first time an octameric resistosome has been identified in plants.
The CCG10-NLR WAI3 resistosome differs from known resistosomes both in the number of monomers and in its conformation, representing a novel assembly mechanism for plant NLR resistosomes.
Using Nicotiana benthamiana and animal cell expression systems, the researchers also demonstrated that the WAI3 resistosome induces Ca2+ influx in plants but is not effective in animal cells. ScienceMission sciencenewshighlights.
Plant immunity is largely initiated at the cellular level, with each cell capable of autonomous detection and response, while also coordinating systemic signaling across the organism—unlike the centralized, cell-based immune system of animals.
One of the key mechanisms of plant immunity involves nucleotide-binding, leucine-rich repeat (NLR) immune receptors—intracellular receptors that detect pathogen invasion and activate a strong immune response. Unlike surface receptors, NLRs detect pathogen effector proteins that are injected by pathogens into plant cells to manipulate host biology.
NLRs are modular proteins with three main parts. They are divided into two main NLR classes—Toll/interleukin-1 receptor-like (TIR) NLRs or coiled-coil (CC) NLRs—based on the structure of their N-terminal domain.