The researchers also discovered a tiny antibody, called a nanobody, which mimics NPC1 at the receptor-binding site and can slip past a protective cap on Marburg’s entry protein, bind to it and block its attachment to the receptor. In lab tests, this nanobody prevented Marburg virus from entering cells. ScienceMission sciencenewshighlights.
In a new study published in Nature the researchers found that the Marburg virus (MBV), one of the world’s deadliest pathogens with an average 73% fatality rate, is unusually efficient at getting inside human cells. They also showed that the virus’s entry protein contains structural features that explain this efficiency and point to a strategy for blocking infection.
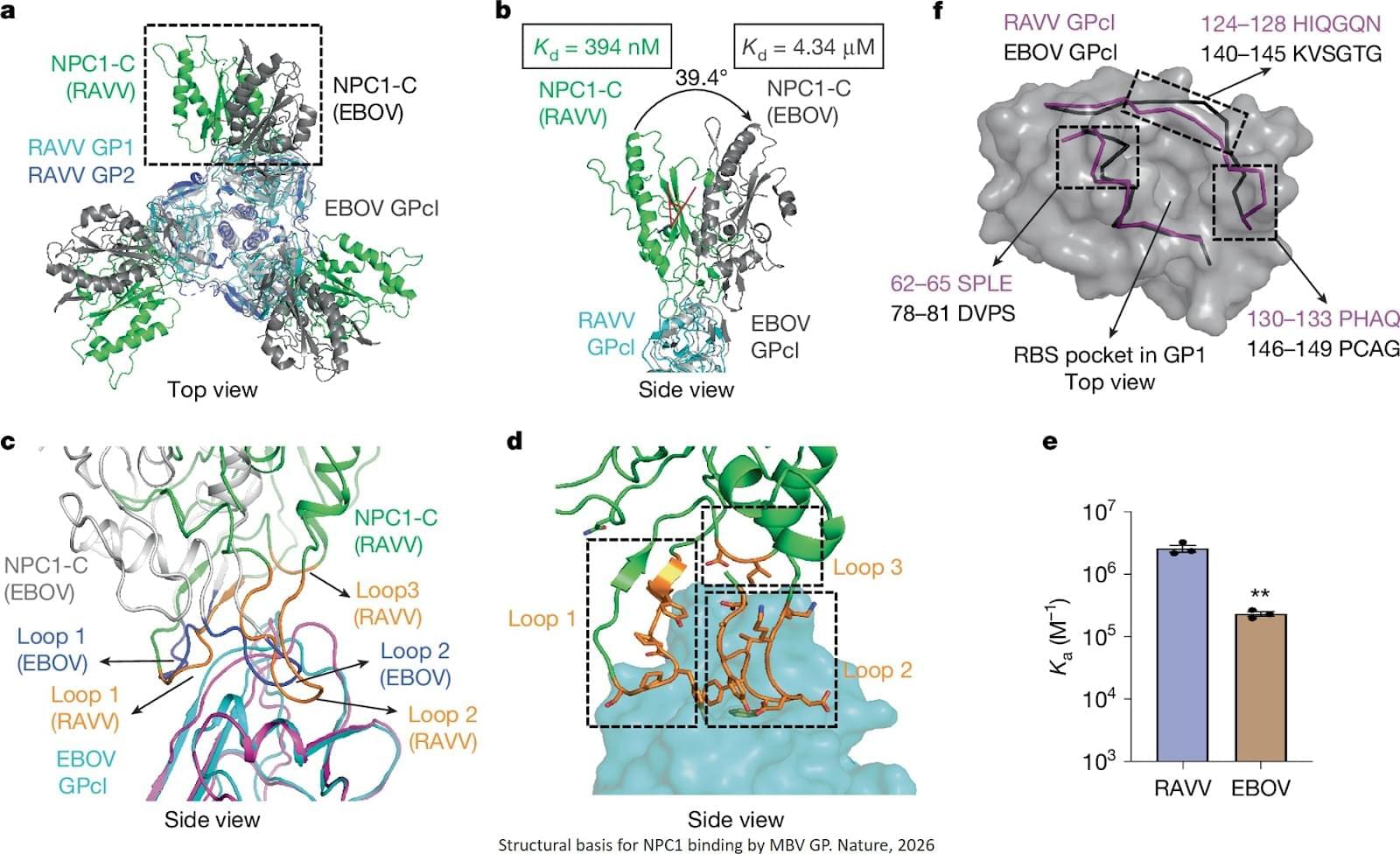
The researchers designed a tightly controlled system that enables a fair comparison of the entry proteins of Marburg and its relative Ebola. The team further found that the two viruses share the same human receptor. The authors determined structures of MBV glycoprotein (GP) in three states: unbound; bound to its endosomal receptor NPC1; and complexed with a neutralizing nanobody.
Marburg’s entry protein binds this receptor in a distinct orientation and with higher affinity, then changes shape in ways that help the virus enter cells. The authors show that the glycan cap shields the receptor-binding site from NPC1 but only partially from the nanobody, enabling limited immune evasion. After glycan cap cleavage, NPC1 binds to MBV GP in a distinct orientation compared with EBOV GP, providing an additional anchor and enhancing receptor affinity. NPC1 engagement also induces substantial conformational changes in MBV GP, probably facilitating membrane fusion. Using this approach, they showed that Marburg’s entry protein can drive viral entry into human cells up to 300 times more efficiently than Ebola’s.