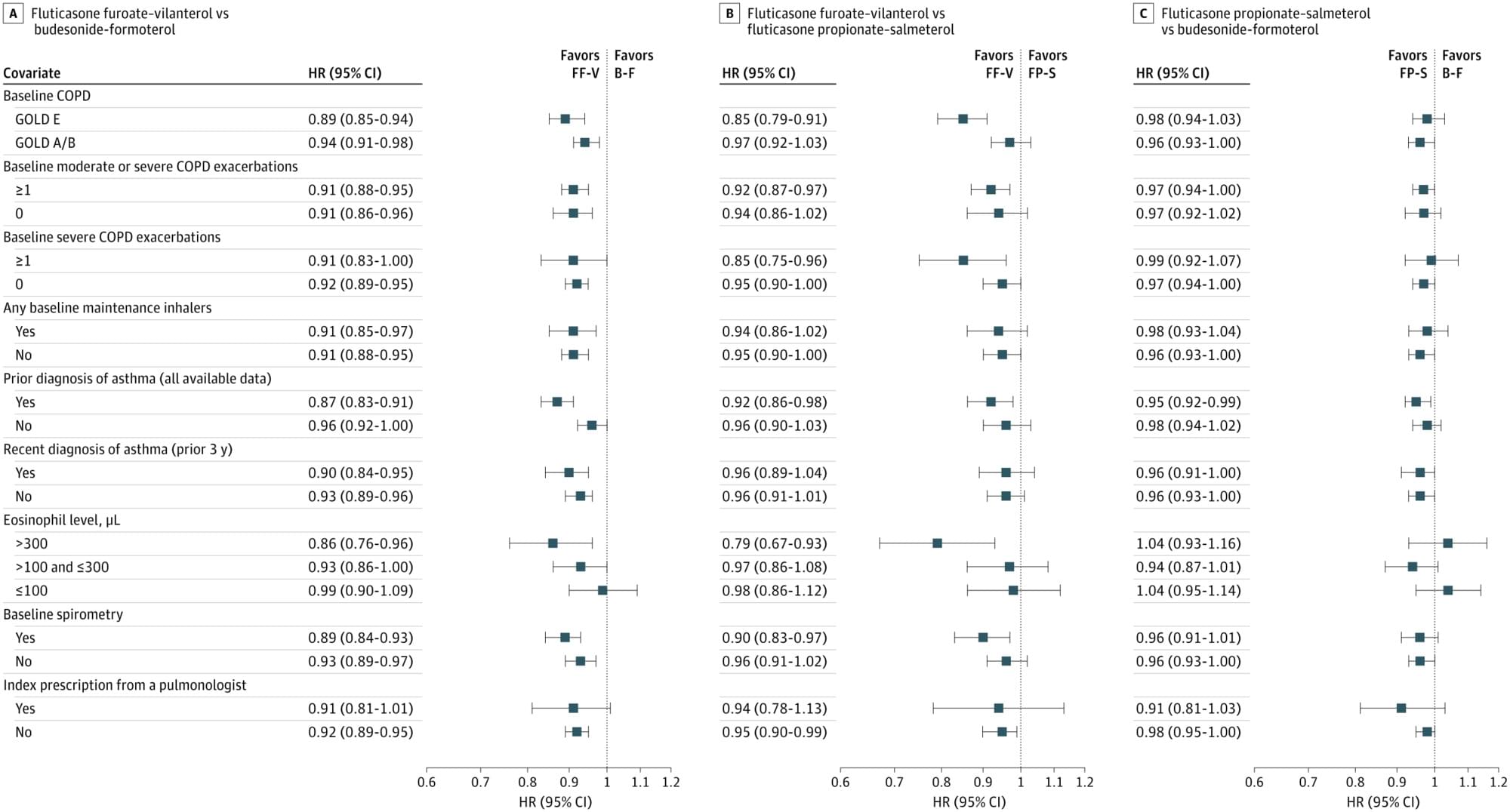
In this cohort study of ICS-LABA therapy in patients with COPD, fluticasone furoate–vilanterol was associated with similar or slightly improved clinical outcomes compared with budesonide-formoterol and fluticasone propionate–salmeterol.
This cohort study was approved by the Mass General Brigham Institutional Review Board. Informed consent was waived because the analysis used deidentified data. The protocol was preregistered with the Center for Open Science (https://osf.io/yp2kh) before the analyses were implemented. The study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.