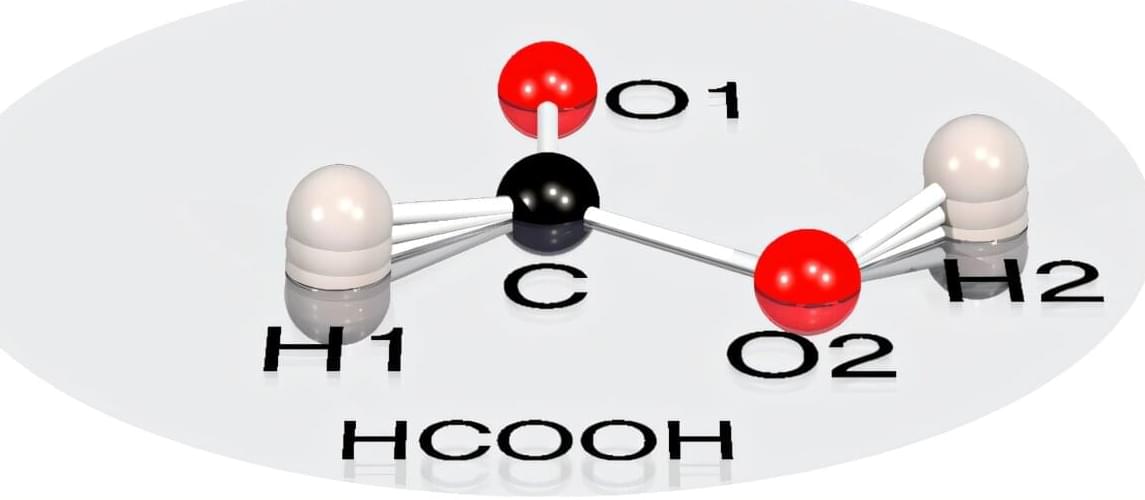
Traditional chemistry textbooks present a tidy picture: Atoms in molecules occupy fixed positions, connected by rigid rods. A molecule such as formic acid (methanoic acid, HCOOH) is imagined as two-dimensional—flat as a sheet of paper. But quantum physics tells a different story. In reality, nature resists rigidity and forces even the simplest structures into the third dimension.
Researchers led by Professor Reinhard Dörner of the Institute for Nuclear Physics at Goethe University have now determined the precise spatial structure of the “flat” formic acid molecule using an X-ray beam from the PETRA III synchrotron radiation source at the DESY accelerator center in Hamburg. They collaborated with colleagues from the universities of Kassel, Marburg and Nevada, the Fritz Haber Institute, and the Max Planck Institute for Nuclear Physics. The study is published in Physical Review Letters.
To accomplish this, they made use of two effects that occur when X-ray radiation strikes a molecule. First, the radiation ejects several electrons from the molecule (photoelectric effect and Auger effect). As a result, the atoms become so highly charged that the molecule bursts apart in an explosion (Coulomb explosion). The scientists succeeded in measuring these processes sequentially, even though they take place within femtoseconds—millionths of a billionth of a second.