To understand the complex interactions involved in an immune response during scarcity, the team put mice on a 50% restricted-calorie diet and then exposed the animals to bacteria that infect the gut. The mice that were fed a standard diet experienced a metabolic crash— their blood glucose levels and body weight plummeted.
The researchers had expected this would happen to all the animals because mounting an immune response can consume up to 30% of the entire body’s fuel reserves. But in the calorie-restricted mice, the immune system appeared to be functioning perfectly well without using much glucose.
To unravel this enigma, the researchers inventoried the immune cells of the infected animals and discovered that T cells, which normally target invading microbes, were depleted in the calorie-restricted mice. Instead, short-lived neutrophils, which serve as the body’s first responders to infection, were ramped up to twice the normal amount and had measurably enhanced pathogen-killing abilities. The cells seemed to be operating in energy-saving mode, consuming much less glucose than neutrophils from well-fed animals.
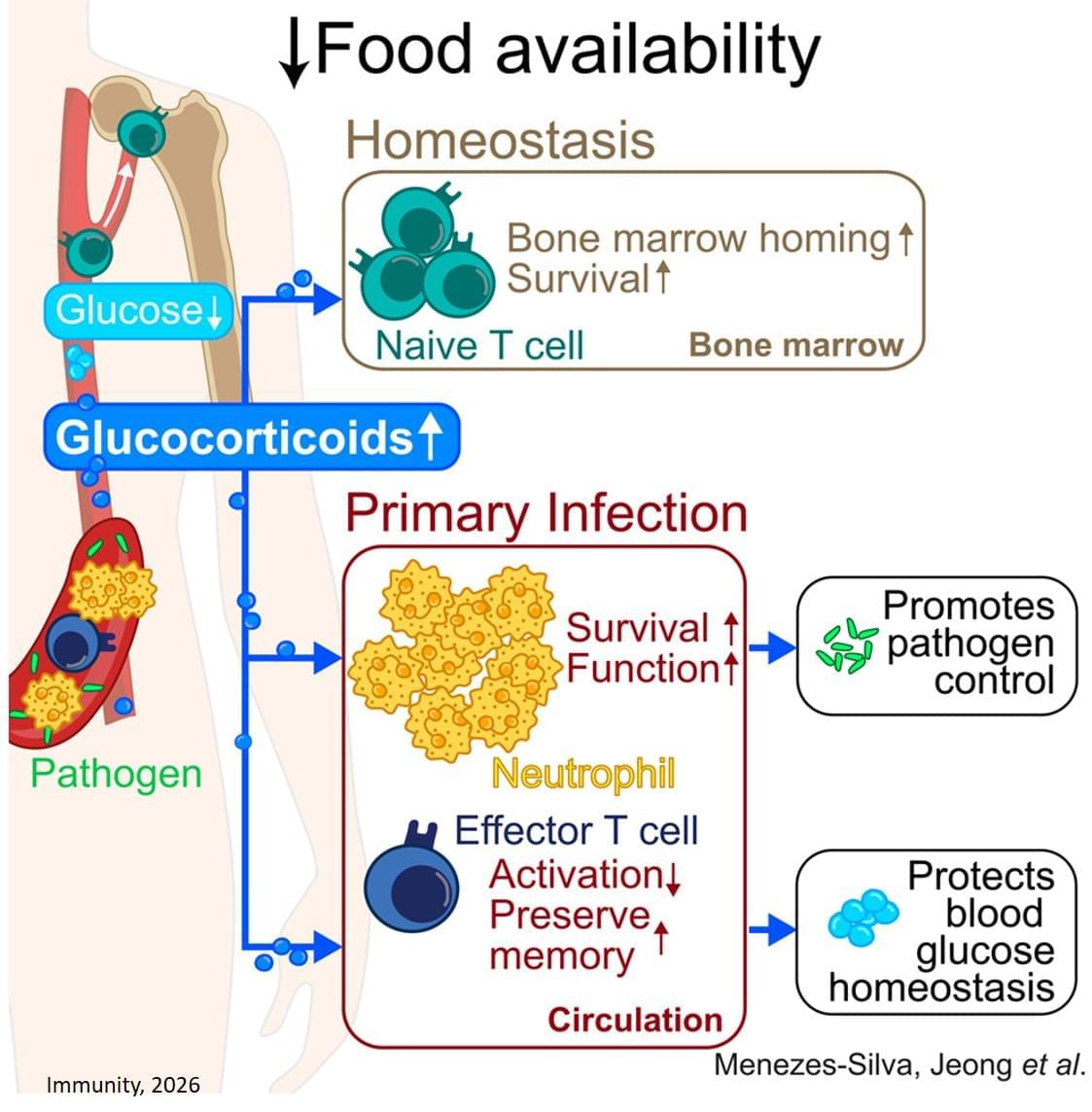
The researchers are breaking new ground by outlining how a sudden fall in food intake triggers glucocorticoid levels to rise, resulting in two major shifts. First, the body repositions certain immune cells—especially naïve T cells—into the bone marrow, which becomes a kind of “safe house” for when the cells are needed. Second, during an infection, glucocorticoids tilt the immune response away from energy-intensive T cells toward neutrophils, abundant cells that act as immediate, short-lived defenders.
Beyond clearing a current infection, glucocorticoids prepare the immune system for repeat encounters with infectious agents. While the hormones direct killer T cells to stand down and neutrophils to step up, they also ensure memory T cells are preserved for future confrontations.
When food is scarce, stress hormones direct the immune system to operate in “low power” mode to preserve immune function while conserving energy, according to researchers. This reconfiguration is crucial to combating infections amid food insecurity.
“Both famine and infectious disease have been with us throughout our evolutionary history and often occurred at the same time. Yet little is known about how nutrition affects the immune system,” said the senior author.
The answer could be important in helping the 47 million Americans who are food insecure and face the risk of infectious diseases every day. “Mounting an immune response against infections requires a lot of energy. We have discovered a coordinated system that upholds immune function by shifting the composition and metabolism of immune cells,” the author said.