Five years of ublituximab treatment in MultipleSclerosis demonstrated sustained reduction in relapse rates and confirmed disability progression, with safety profile consistent over time.
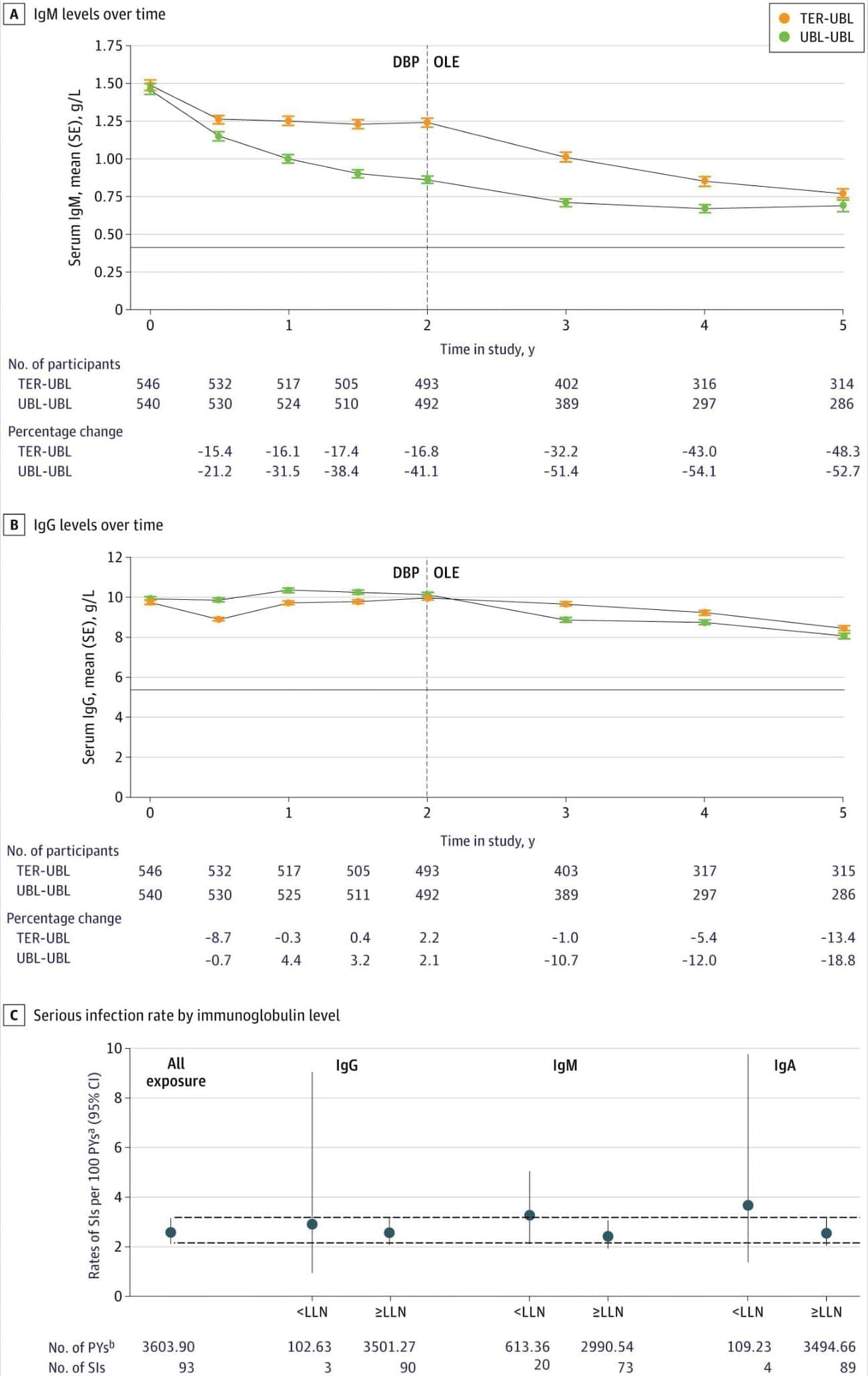
Question What is the long-term clinical efficacy and safety of ublituximab in people with relapsing multiple sclerosis (RMS)?
Findings In this trial including 985 adults, participants treated with continuous ublituximab for up to 5 years in the open-label extension study after completion of the randomized Study to Assess the Efficacy and Safety of Ublituximab in Participants With Relapsing Forms of Multiple Sclerosis (ULTIMATE) had significantly lower annualized relapse rate and confirmed disability progression than those initially treated with teriflunomide. The overall safety profile of ublituximab remained consistent with no new safety signals emerging with prolonged treatment.
Meaning Results suggest that early initiation of ublituximab and continued treatment over a period of 5 years provided sustained clinical benefits in participants with RMS.