For a phenomenon like a wildfire burning through a forest or a disease moving through a population, the resulting patterns can sometimes be modeled using a so-called reaction–diffusion system—an experiment where a chemical reaction front moves through a region full of reactants. Now Anne De Wit of the Université Libre de Bruxelles and her colleagues have demonstrated that new patterns can be revealed when the reactants flow against the direction of the front’s propagation, causing it to freeze in place [1]. Their “sun-ray” pattern is the first one discovered this way, but the technique could generate other patterns that might replicate behavior in forest fires or epidemics.
Two years ago, De Wit and her colleagues brought a propagating reaction front to a standstill by slowly and continually injecting a reactant into the center of a disk-shaped chamber filled with the other reactant, against the front’s inward propagation [2]. The stopping occurred when the outward flow matched the rate at which the inner reactant was consumed. De Wit says that a stationary front allows more control and thus more careful study of patterns than a propagating front.
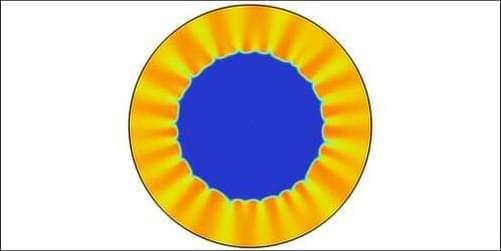
As a demonstration of this control, the researchers have now used reactants with different diffusion rates in the same outward-flow setup. In this case, the stationary front developed ripples—an effect previously seen in propagating fronts. The researchers also observed radial “rays”—narrow regions of higher concentration of one of the reactants. They showed in simulations and experiments that properties of the front can be precisely controlled by varying the flow rate.