Controlling mucocutaneous barriers by cytokines.
Impaired IL-17 leads to mucocutaneous bacterial and fungal infections, whereas enhanced IL-17 promotes psoriasis and periodontitis.
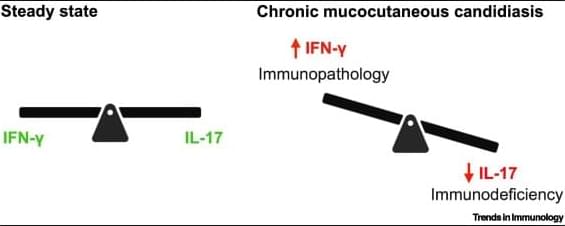
Interferon (IFN)-γ deficiency causes infections by intracellular pathogens, whereas exacerbated mucosal IFN-γ activity promotes oral candidiasis, even when IL-17 responses are intact.
JAK inhibition in autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy remits oral candidiasis and multiorgan autoimmunity.
IFN-γ drives Candida auris skin infection by impairing the epithelial barrier. Excess IFN-γ, tumor necrosis factor, and types I/III IFN activities contribute to pathology during bacterial and protozoan skin infections and pulmonary viral, bacterial, mycobacterial, and fungal infections.
Although immune deficiency causes mucosal infection, epithelial disruption by immunopathology represents a novel and underappreciated mechanism of infection susceptibility at barrier sites. sciencenewshighlights ScienceMission https://sciencemission.com/Balancing-IL-17
Mucocutaneous surfaces rely on IL-17–producing lymphocytes to preserve barrier integrity and prevent bacterial and fungal overgrowth. Accordingly, genetic or pharmacological IL-17 deficiencies lead to mucocutaneous infections. Interferon (IFN)-γ mediates host defense against intracellular pathogens, but excessive mucosal IFN-γ activity can paradoxically impair epithelial integrity and promote infection, as shown in autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy–associated oral candidiasis, even with intact IL-17 responses. Further evidence for IFN-γ–driven pathology is emerging in bacterial, fungal, and protozoal infections at mucocutaneous tissues. Together, these findings support a model in which IL-17 promotes barrier resistance, whereas unchecked IFN-γ erodes it.