After years of trying, scientists have finally created a stable superatom of copper, a long-sought-after chemical breakthrough that could revolutionize how we deal with carbon emissions.
Copper is a cheap and common metal, and because of its ability to bind carbon atoms together (C-C coupling), scientists have wanted to use it to turn carbon dioxide into products like ethylene for plastics and fuels. However, it corrodes or falls apart almost immediately when exposed to air or harsh industrial conditions.
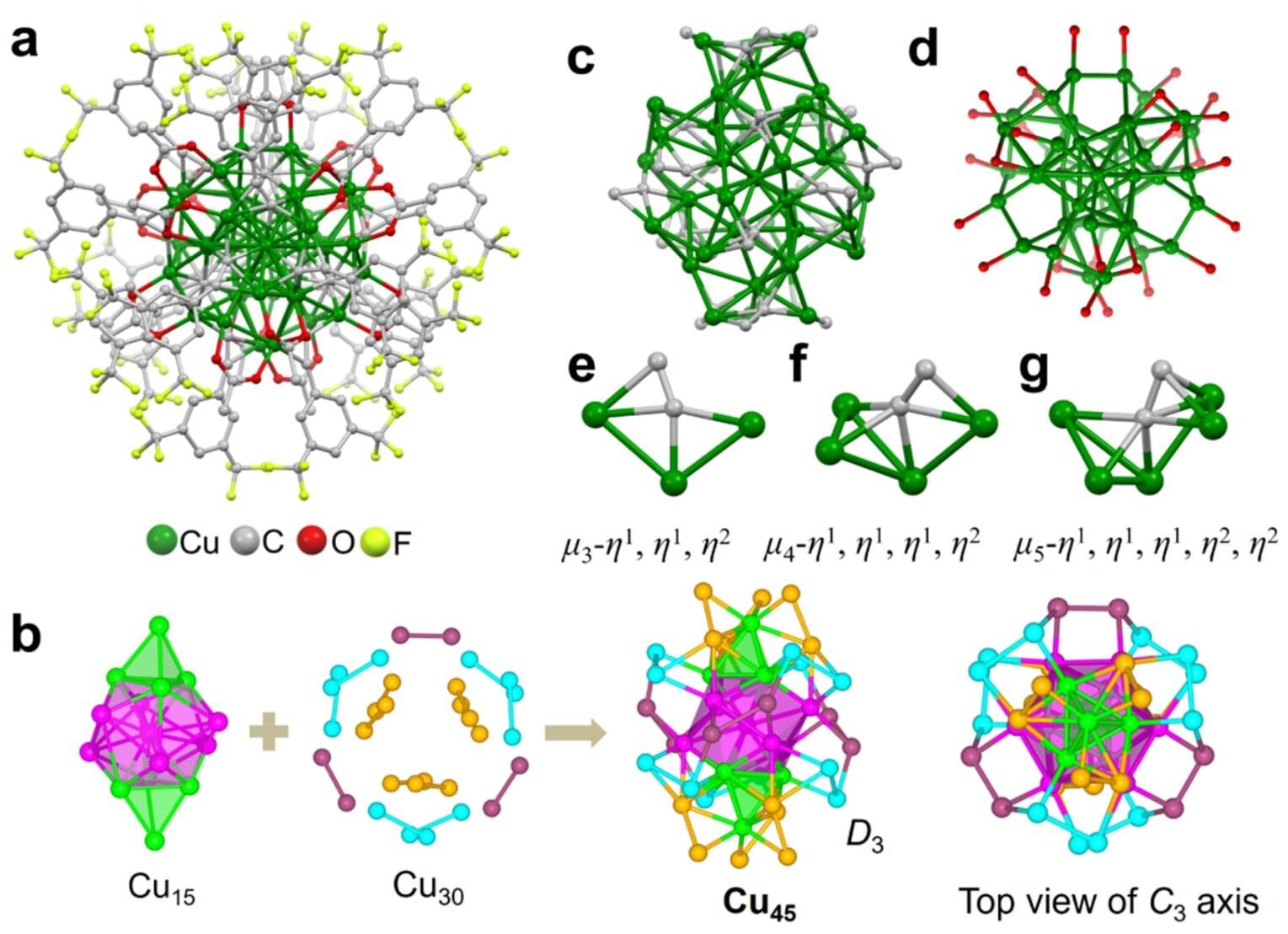
A superatom is a cluster of atoms that behaves like a single atom, but with greater stability. In this new study published in the Journal of the American Chemical Society, scientists from Tsinghua University in Beijing built a nanocluster made from 45 copper atoms (Cu45).