📝 — Sheikh, et al.
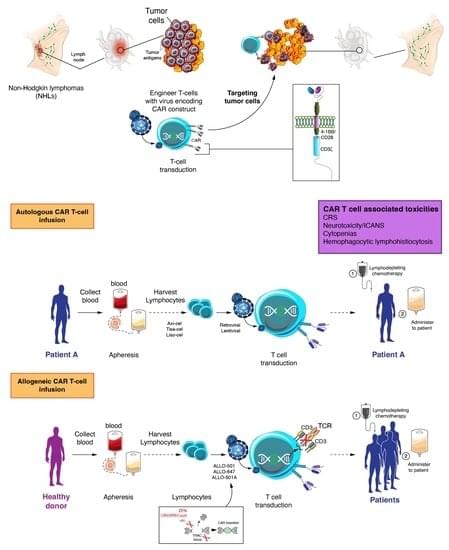
In this paper, the authors summarize the current practice and the latest progress of CD19 auto-CAR T cell therapy and the management of specific toxicities and discuss the place of allogeneic CAR T development in this setting.
Full text is available 👇
While more than half of non-Hodgkin lymphomas (NHL) can be cured with modern frontline chemoimmunotherapy regimens, outcomes of relapsed and/or refractory (r/r) disease in subsequent lines remain poor, particularly if considered ineligible for hematopoietic stem cell transplantation. Hence, r/r NHLs represent a population with a high unmet medical need. This therapeutic gap has been partially filled by adoptive immunotherapy. CD19-directed autologous chimeric antigen receptor (auto-CAR) T cells have been transformative in the treatment of patients with r/r B cell malignancies. Remarkable response rates and prolonged remissions have been achieved in this setting, leading to regulatory approval from the U.S. Food and Drug Administration (FDA) of four CAR T cell products between 2017 and 2021.