Novel study designed to correct genetic abnormalities of red blood cells.
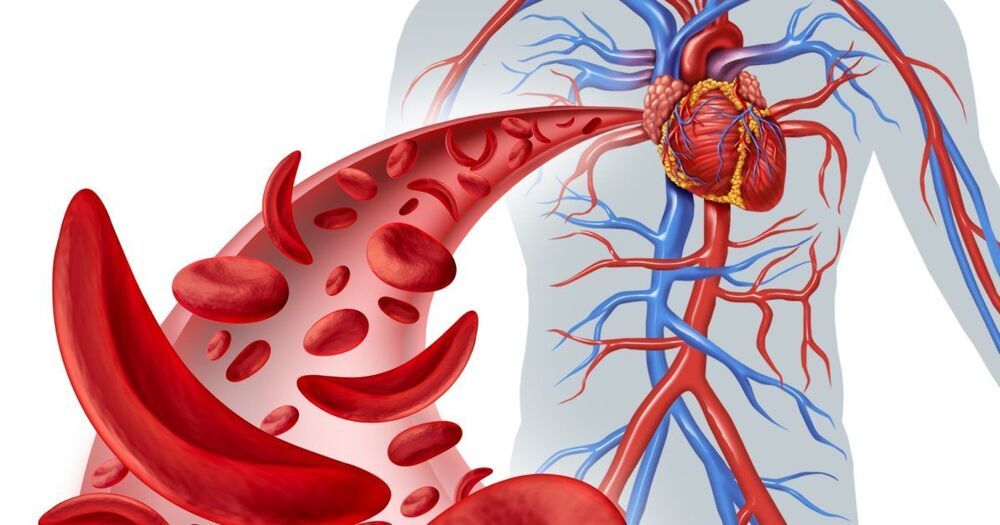
Cleveland Clinic researchers are enrolling patients in a clinical trial that aims to work toward a cure for sickle cell disease, by changing the patient’s genetics. Sickle cell disease, a genetic blood disorder, is a painful and debilitating condition for which there are few approved therapies.
The multicenter study will evaluate the safety and effectiveness of a single dose of EDIT-301, an experimental one-time gene editing cell therapy that modifies a patient’s own blood-forming stem cells to correct the mutation responsible for sickle cell disease.
During the study patients’ stem cells are collected for gene editing in a laboratory. Patients then are treated with chemotherapy to destroy remaining bone marrow to make room for the repaired cells which are infused back into the body. The study will initially enroll 40 adult patients ages 18 to 50 with severe sickle cell disease, with the possibility of expansion to include adolescents. Patients will be monitored closely after treatment for up to two years.