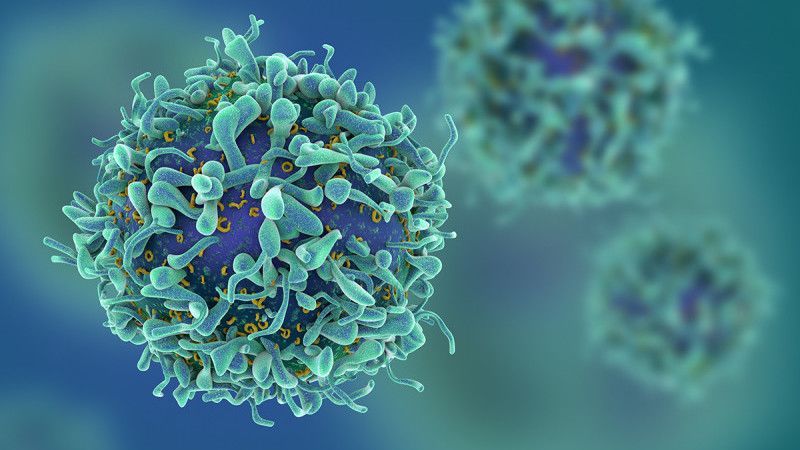
On Wednesday—for only the second time—the Food and Drug Administration approved a cutting-edge therapy that genetically modifies a patient’s blood cells in order to attack cancer. This time the therapy, known as CAR T-cell therapy, is designed to treat aggressive non-Hodgkin lymphoma.
In August, the FDA approved the first CAR T-cell therapy, for a drug called Kymriah designed for children and young adults whose leukemia doesn’t respond to standard treatments. The FDA’s approval of Yescarta, manufactured by Kite Pharma, comes just a few months after its first approval—an indication of just how quickly the field of immunotherapy is moving. Several other companies also have CAR-T therapies in the works.