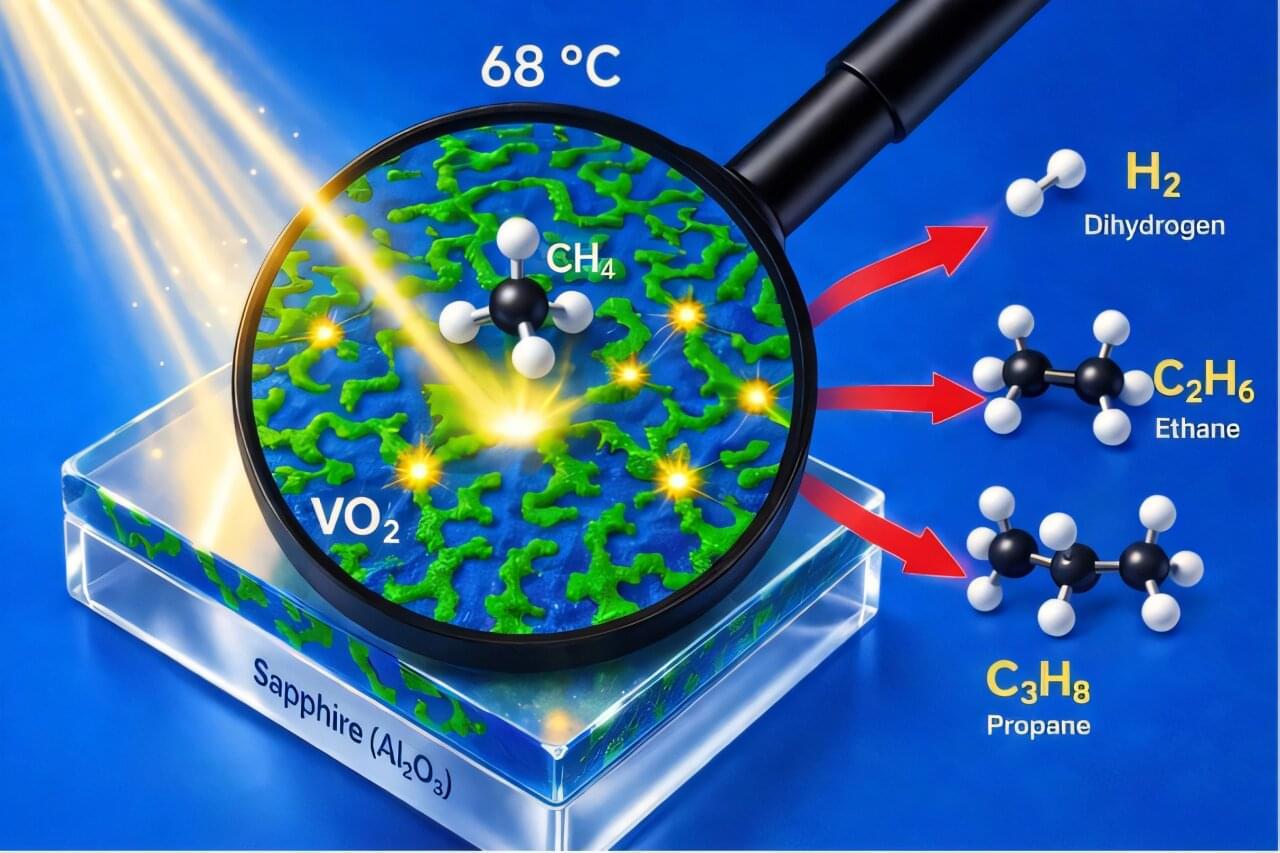
Converting methane, the primary component of natural gas, into higher alkanes and hydrogen, could be highly advantageous. Alkanes, such as propane and butane, are easier to transport than methane and are used in a wider range of industries. Hydrogen, on the other hand, is a promising clean fuel used to power electrochemical devices that can generate continuous power, known as fuel cells.
Over the past decades, some energy engineers have been exploring the possibility of converting methane into hydrogen or complex hydrocarbons using photocatalysts. These are materials activated by sunlight or other types of light and that can drive chemical reactions.
Researchers at Université de Lille—CNRS, Sorbonne Université and other institutes in France recently introduced a new strategy for the photocatalytic conversion of methane into propane, which is widely used for heating, cooking, and transportation.