Columnist Natalie Wolchover examines the latest developments in the “forever war” over whether string theory can describe the world.
Get the latest international news and world events from around the world.

Age Reversal 2026: Why Getting Old Will Soon Be Optional
Is aging actually optional? According to this Harvard scientist, yes—and human trials start soon. He explains why aging is not inevitable but a medical condition we can now treat, sharing groundbreaking results from his lab including reversing blindness in animals and rejuvenating biological age by 75% in just six weeks.
The FDA has just approved the first human trials for age reversal, marking a turning point in medical history. This video covers the science, the economics, and a future where we spend our 80s and 90s as healthy as our 40s. This is the update you’ve been waiting for.
Credits to World Governments Summit & Dr David Sinclair
Please note that the links below are affiliate links, so we receive a small commission when you purchase a product through the links. Thank you for your support!
=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=*=
📒N1O1 10% OFF with Code : REVERSE 📒
Nitric Oxide Booster https://tinyurl.com/4krxe2tx.
🔶 Nuchido https://www.nuchido.com/REVERSEAGING FIRST ORDER 20% OFF code : REVERSEAGING20
🆕 Discount Coupon Code : REVERSE — ProHealth, RENUE, StemRegen, N1O1, NOVOS ☑
🔲 Activate Your Stem Cells Naturally — STEMREGEN 15% OFF https://tinyurl.com/StemRelease.
🌏ProHealth Longevity15% OFF, Subscription Upto 45% OFF Use Code “REVERSE”
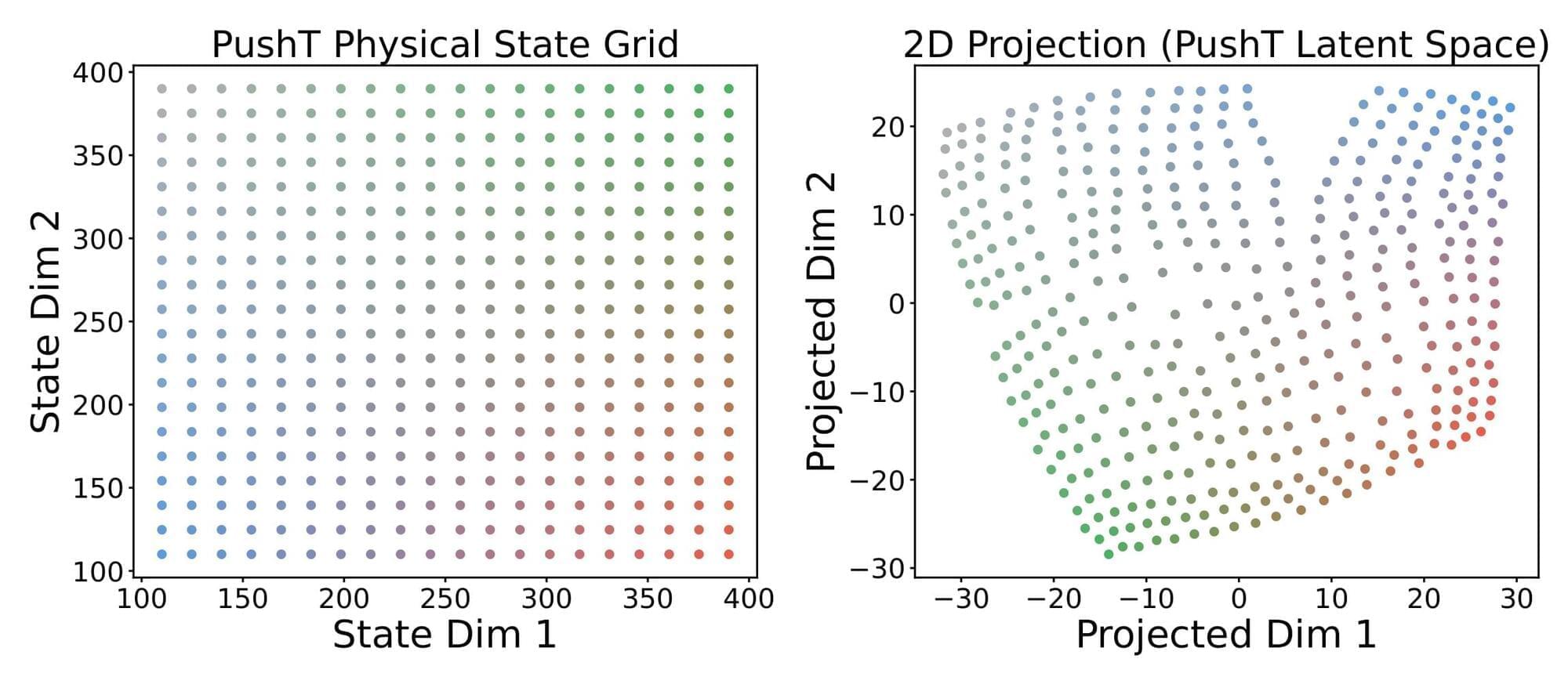
LeWorldModel: Stable End-to-End Joint-Embedding
Joint Embedding Predictive Architectures (JEPAs) offer a compelling framework for learning world models in compact latent spaces, yet existing methods remain fragile, relying on complex multi-term losses, exponential moving averages, pre-trained encoders, or auxiliary supervision to avoid representation collapse. In this work, we introduce LeWorldModel (LeWM), the first JEPA that trains stably end-to-end from raw pixels using only two loss terms: a next-embedding prediction loss and a regularizer enforcing Gaussian-distributed latent embeddings. This reduces tunable loss hyperparameters from six to one compared to the only existing end-to-end alternative. With ~15M parameters trainable on a single GPU in a few hours, LeWM plans up to 48× faster than foundation-model-based world models while remaining competitive across diverse 2D and 3D control tasks. Beyond control, we show that LeWM’s latent space encodes meaningful physical structure through probing of physical quantities. Surprise evaluation confirms that the model reliably detects physically implausible events.
TL;DR: LeWM is a JEPA-based world model that avoids representation collapse using a simple Gaussian regularizer (SIGReg), trains end-to-end from pixels with only two loss terms, and achieves competitive control performance at a fraction of the compute cost.
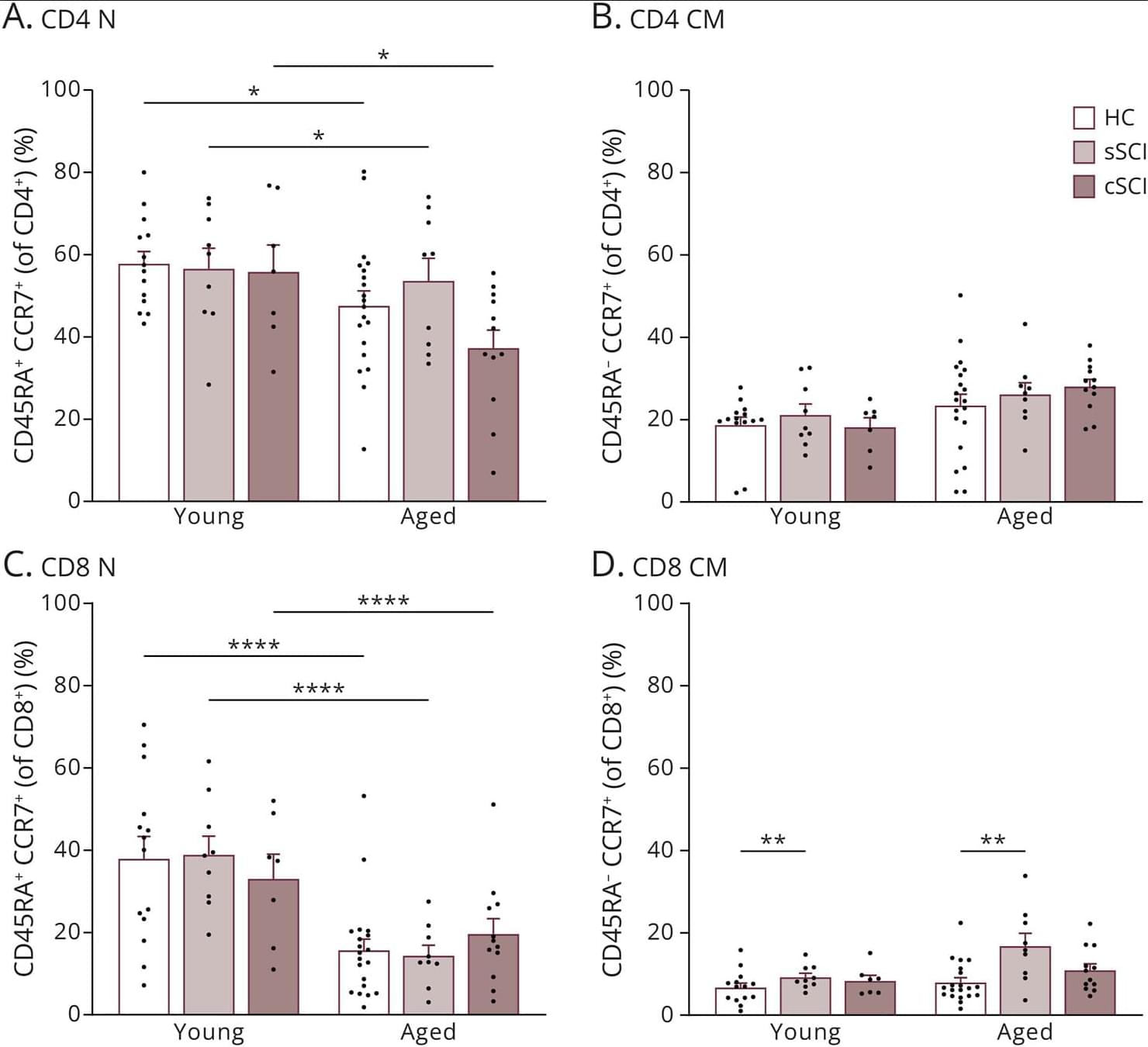
Peripheral Immune Remodeling After Spinal Cord InjuryMarked by Expansion of CD38+, CTLA-4+, and PD-1+ T and NK Cells
Study findings reveal noteworthy changes in immune cell activation and exhaustion markers that may contribute to immune vulnerability after spinal cord injury, offering novel insights into potential therapeutic targets, such as NAD+ metabolism and immune checkpoint modulation.
Background and Objectives.
The Biggest Threat to Cosmology
The Big Bang just doesn’t make much sense. We don’t have any real idea what was before (or rather ‘beyond’) it, but some of our best guesses involve quantum fluctuations and chance. While this makes intuitive sense, it leads to some statistical consequences #science #bigbang #quantum #boltzmannbrain …
Rocky Is Weirder Than You Think (ft. Andy Weir!)
Go to http://incogni.com/joescott and you can get 60% off an annual Incogni plan that protects your private info online.
Project Hail Mary opened in theaters last week, introducing the world to Rocky, the alien creature whose friendship with the main character, Ryland Grace, forms the heart of the story. Rocky quickly became a fan favorite of readers of the book, partly because of the extensive research and imagination put into the creature by the author, Andy Weir. In today’s video, I have Andy Weir join me to break down everything you could possibly want to know about Rocky, from his (not so) fictional planet to his crazy anatomy. It’s a masterclass in speculative biology that will amaze amaze amaze you.
Check out the Oldest and Newest Places posters, now on sale!
https://laughsmarter.com/collections/.… to support the channel? Here’s how: Patreon: / answerswithjoe Channel Memberships:
/ @joescott T-Shirts & Merch: https://laughsmarter.com Book of Mysteries: https://a.co/d/0gRx0qvM Documentary: https://nebula.tv/oldestnewest Check out my 2nd channel, Joe Scott TMI:
/ @joescott-tmi And my podcast channel, Conversations With Joe:
/ @conversationswithjoe You can listen to my podcast, Conversations With Joe on Spotify, Apple Podcasts, Google Podcasts, or wherever you get your podcasts. Spotify 👉 https://spoti.fi/37iPGzF Apple Podcasts 👉 https://apple.co/3j94kfq Google Podcasts 👉 https://bit.ly/3qZCo1V Follow me at all my places! Instagram:
/ answerswithjoe TikTok:
/ answerswithjoe Facebook:
/ answerswithjoe Twitter:
/ answerswithjoe TIMESTAMPS 0:00 — Intro 3:15 — About The Eridani System 7:10 — Morphology 18:04 — Crystal Brain 20:44 — Digestion 24:19 — Circulation/Musculature/Dormancy 28:37 — Communication 35:00 — How They Brought Rocky to Life 38:16 — Sponsor — Incogni.
Want to support the channel? Here’s how:
Patreon: / answerswithjoe.
Channel Memberships: / @joescott.
T-Shirts & Merch: https://laughsmarter.com.
Book of Mysteries: https://a.co/d/0gRx0qvM
Documentary: https://nebula.tv/oldestnewest.
Check out my 2nd channel, Joe Scott TMI:
The 12 Most Distant Places Ever Reached in Star Trek
What is the farthest place ever reached in Star Trek?From dangerous nebulae just a few hundred light-years away… to the literal edge of the observable univer…

Next-generation memory material has the surprising property of shrinking when heated
Most materials we use in everyday life expand slightly when heated and return to their original size when cooled. In addition to such thermal properties, materials can also have electrical properties or magnetic properties, and traditionally we have used these characteristics separately. However, some materials allow multiple properties to coexist within a single substance.
Research on such materials is expected to contribute to the development of next-generation memory devices that can store and retain information while consuming far less energy.
How multiferroics could transform memory A representative example is a class of materials known as multiferroics, which combine the properties of a capacitor (the ability to store electric charge) and a magnet. Among them, bismuth ferrite (BiFeO₃) is one of the most intensively studied materials in the field. When an external voltage is applied, the direction of its stored electric polarization can be switched, and this change can also influence its magnetic properties.
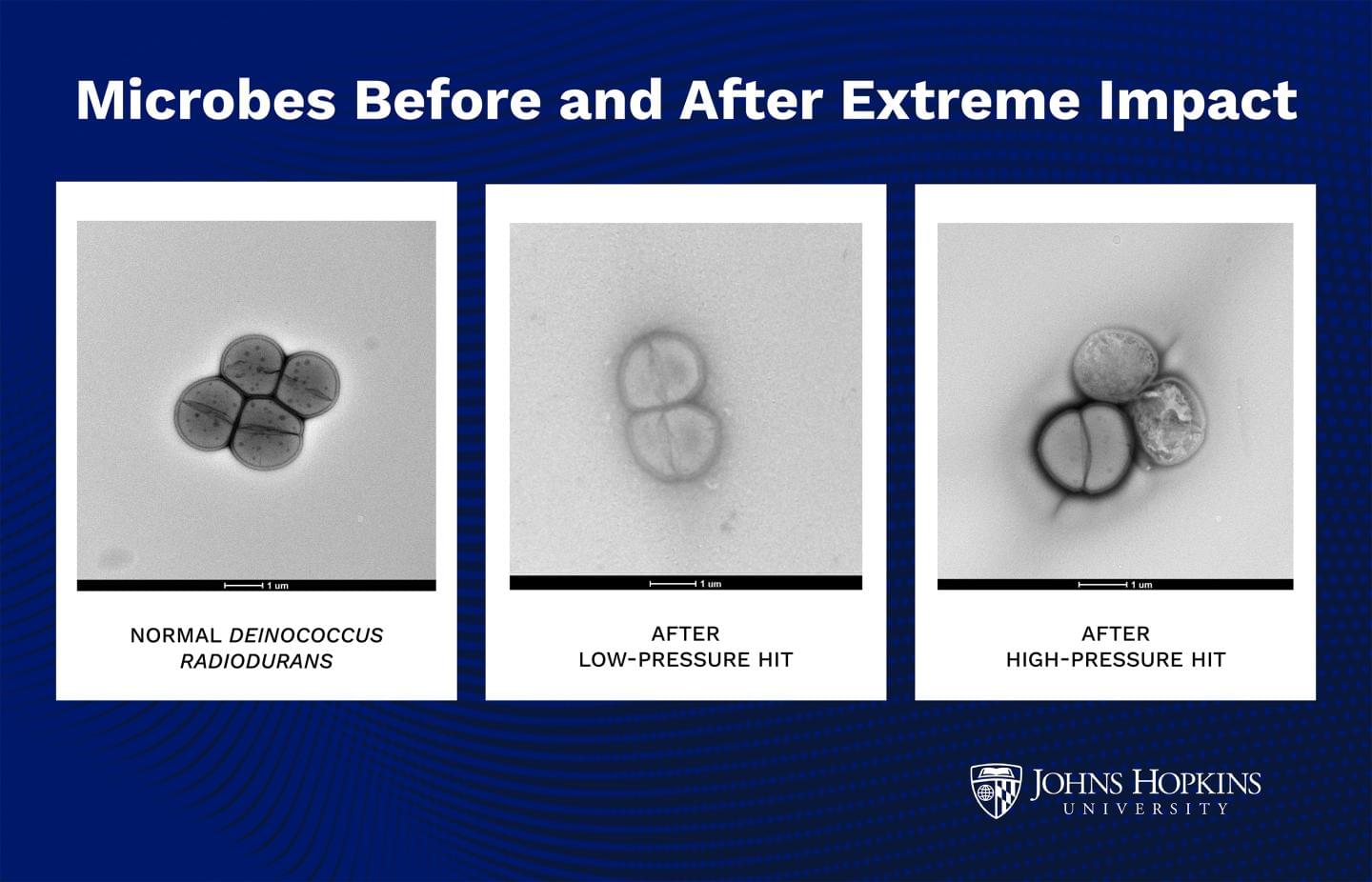
Life forms can planet hop on asteroid debris—and survive
Tiny life forms tucked into debris from an asteroid hit could catapult to other planets—including Earth—and survive, a new Johns Hopkins University study finds. The work demonstrates that a certain hardy bacterium easily withstands extreme pressure comparable to an ejection from Mars after an asteroid hit, as well as the inhospitable conditions it would face during the ensuing interplanetary journey.
The study, published today in PNAS Nexus, suggests that microorganisms can survive remarkably more extreme conditions than expected, and raises questions about origins of life. The work also has significant implications for planetary protection and space missions.
“Life might actually survive being ejected from one planet and moving to another,” said senior author K.T. Ramesh. “This is a really big deal that changes the way you think about the question of how life begins and how life began on Earth.”