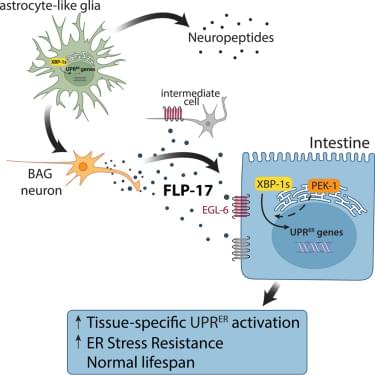
FLP-17’s role in stress resistance aligns with its established functions. FLP-17 belongs to an evolutionarily conserved class, FMRF-amide/RF-amide neuropeptides, that plays important roles in energy balance and reproduction across phyla.34,35 In C. elegans, FLP-17 is secreted from a pair of sensory neurons (BAG) in response to low oxygen and high carbon dioxide, which can be caused by unfavorable food conditions or pathogens.36,37 FLP-17 then acts through specific neurons to inhibit egg laying and initiate an aversion behavior until the animal has reached more favorable conditions.30,36 Interestingly, unfavorable food conditions and pathogens also threaten organismal protein homeostasis.33,38 Therefore, we speculate that FLP-17 evolved to simultaneously protect the animal from proteotoxic stress while facilitating a behavioral program to help the animal navigate to more favorable conditions.
To coordinate adaptive behavioral and metabolic responses, FLP-17 primarily signals through the GPCR EGL-6 in specific neurons.30,31 Therefore, we tested whether EGL-6 also mediates FLP-17’s role in UPRER activation and found that FLP-17-induced activation of the UPRER and ER stress resistance is partially dependent on EGL-6. Egl-6 expression is predominantly neuronal, evidenced by transcriptional reporters and single-cell RNA-seq datasets.30,39 However, low levels of egl-6 expression were detected in intestine-specific translation of ribosome-affinity purification, which may better reflect protein levels.40 This suggests that FLP-17 may signal either through an intermediate cell type (such as a neuron) or directly to the intestine to activate UPRER.30,39 Furthermore, the partial dependence, combined with persistent stress gene activation in egl-6 (lof) backgrounds (Figure 5 E), indicates that additional unidentified receptors and mechanisms likely contribute to FLP-17 phenotypes.
Although FLP-17 was sufficient to activate the UPRER, it was not required for cell non-autonomous activation of the UPRER by glial:: xbp-1s, as flp-17 null mutants did not suppress glial:: xbp-1s phenotypes. This likely reflects neuropeptide network redundancy. Supporting this hypothesis, flp-17 (lof)) resulted in modest upregulation of stress response genes (Figure S3G) and a slight increase in hsp-4p::GFP in the glial:: xbp-1s animals (Figure 2D), suggesting compensatory activation of stress signaling pathways when FLP-17 is absent. This compensation could occur through multiple mechanisms. First, glial:: xbp-1s may induce multiple neuropeptides that provide functionally redundant UPRER activation. While no other candidate from our neuropeptidomics screen was individually sufficient to induce UPRER, we cannot exclude compensation by peptides not detected in our analysis, such as insulin-like peptides.