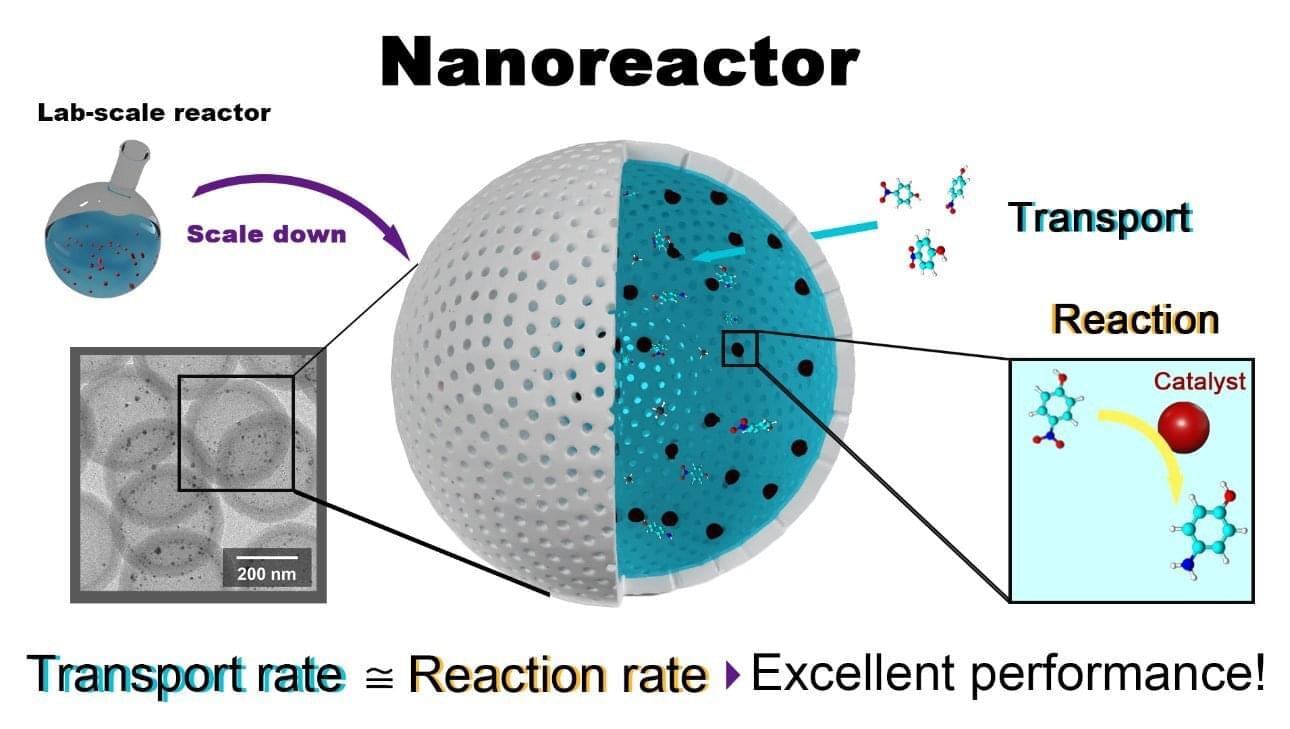
A new study by a team at Tohoku University, published in Chemical Engineering Journal, has shown that more isn’t always better when it comes to nanoscale chemical reactions. One might think that giving reactants completely unrestricted access to a speed-boosting catalyst would be the fastest way to drive a chemical reaction. Instead, it was shown that hollow nanoreactors can work more efficiently when transport into the reaction space is slightly restricted.
A nanoreactor is a porous shell that surrounds an inner space containing catalytically active nanoparticles. The inner space where reactions occur provides a special environment which opens the door for unique and highly useful chemical reactions. Finding ways to optimize reactions in these confined spaces could help to produce a myriad of everyday products more efficiently, and at a lower price.
While it might seem like flooding this inner space would get things done the fastest, researchers found that the key to optimization involved holding back a little.