The transitions of hydrogen molecules embedded in a crystal depend on the surroundings—a behavior that could be used to tailor molecular quantum dynamics.
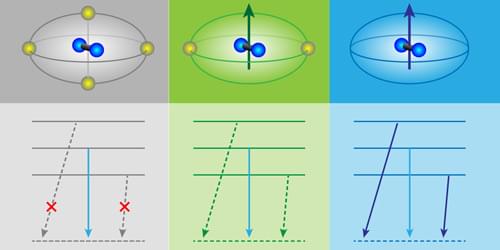
In quantum physics, we often learn that the rules governing a system are set by its symmetry. These rules—known as selection rules—determine which transitions between quantum states are allowed and which are forbidden. For example, rotational symmetry constrains how an atom’s angular momentum can change. But what if those rules are not fixed? A recent study of hydrogen (H2)—one of the simplest molecules in nature—showed that the allowed pathways between quantum states are determined not solely by the molecule’s internal symmetry but also by its surroundings. By embedding hydrogen molecules in different crystalline environments, Nathan McLane and colleagues from the University of Maryland, College Park, have demonstrated that the symmetry of the host material can selectively enable or suppress nuclear-spin transitions [1]. In doing so, the team revealed that quantum dynamics is not just an intrinsic property—it can be shaped by the environment.
H2 is one of the simplest systems for exploring quantum behavior. Its two identical protons can align their spins in two different ways: In so-called orthohydrogen the nuclear spins are parallel, whereas in parahydrogen they are antiparallel. Although this difference is subtle, it leads to markedly different physical properties for the two forms. Crucially, transitions between them are highly constrained: In an isolated hydrogen molecule, the overall wave function is symmetric under exchange of the two protons, and this exchange symmetry forbids direct conversion between ortho and para states [2]. This restriction makes H2 a textbook example of how symmetry governs quantum dynamics.