Urea is an extremely important chemical, especially for fertilizers. But, making urea is energy intensive and relies heavily on fossil fuels. However, new findings from Griffith University and the Queensland University of Technology have highlighted new ways to produce urea electrochemically, using electricity and waste gases such as carbon monoxide (CO) and nitrogen oxides (NO) instead.
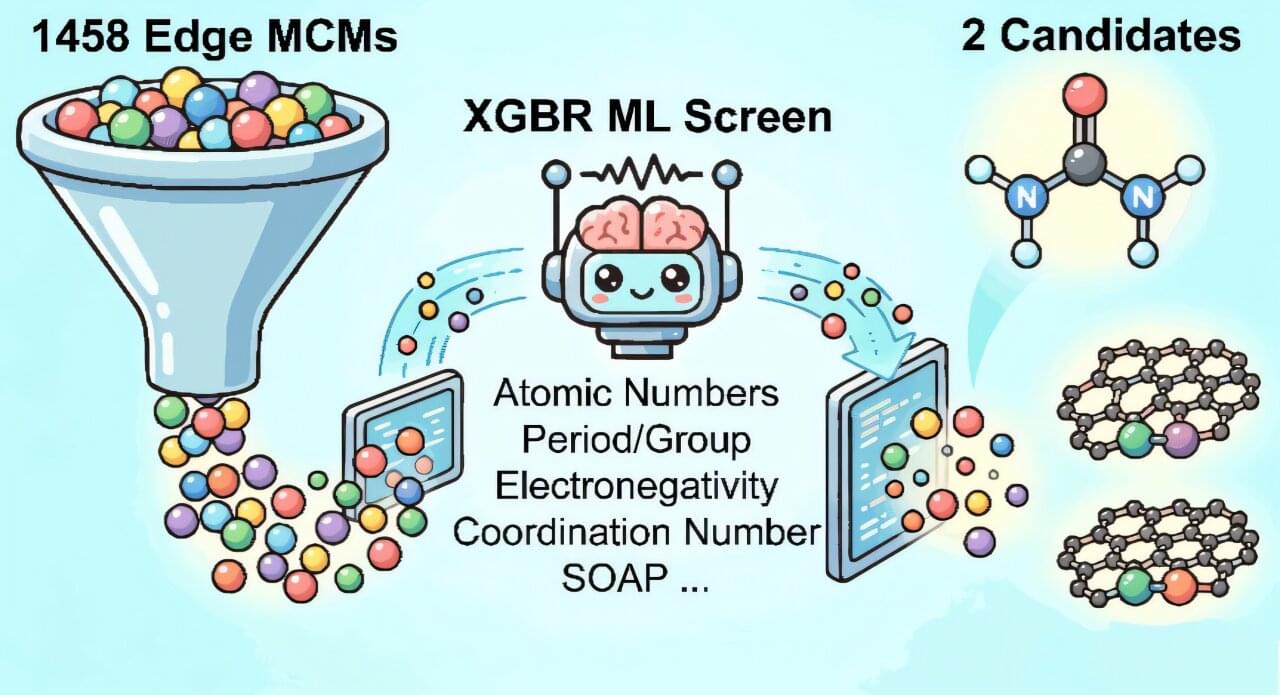
The paper, “Machine Learning-Assisted Design Framework of Carbon Edge-Dominated Dual-Atom Catalysts for Urea Electrosynthesis,” has been published in ASC Nano.
“The challenge is that when CO and NO react on a catalyst, they usually don’t form urea,” said co-lead author Professor Qin Li from Griffith University.