Iridium oxide is one of the most important—and most problematic—materials in the global push toward clean energy. It is currently the most reliable catalyst used in the conversion of energy to chemicals by electrolysis, a process that uses electricity to split water molecules into oxygen and hydrogen.
But iridium is among the rarest non-radioactive elements in Earth’s crust, and not unlike metal rusting over time, iridium oxide catalysts slowly degrade under the harsh acidic and high-voltage conditions required for electrolyzers (the devices used for electrolysis) to operate.
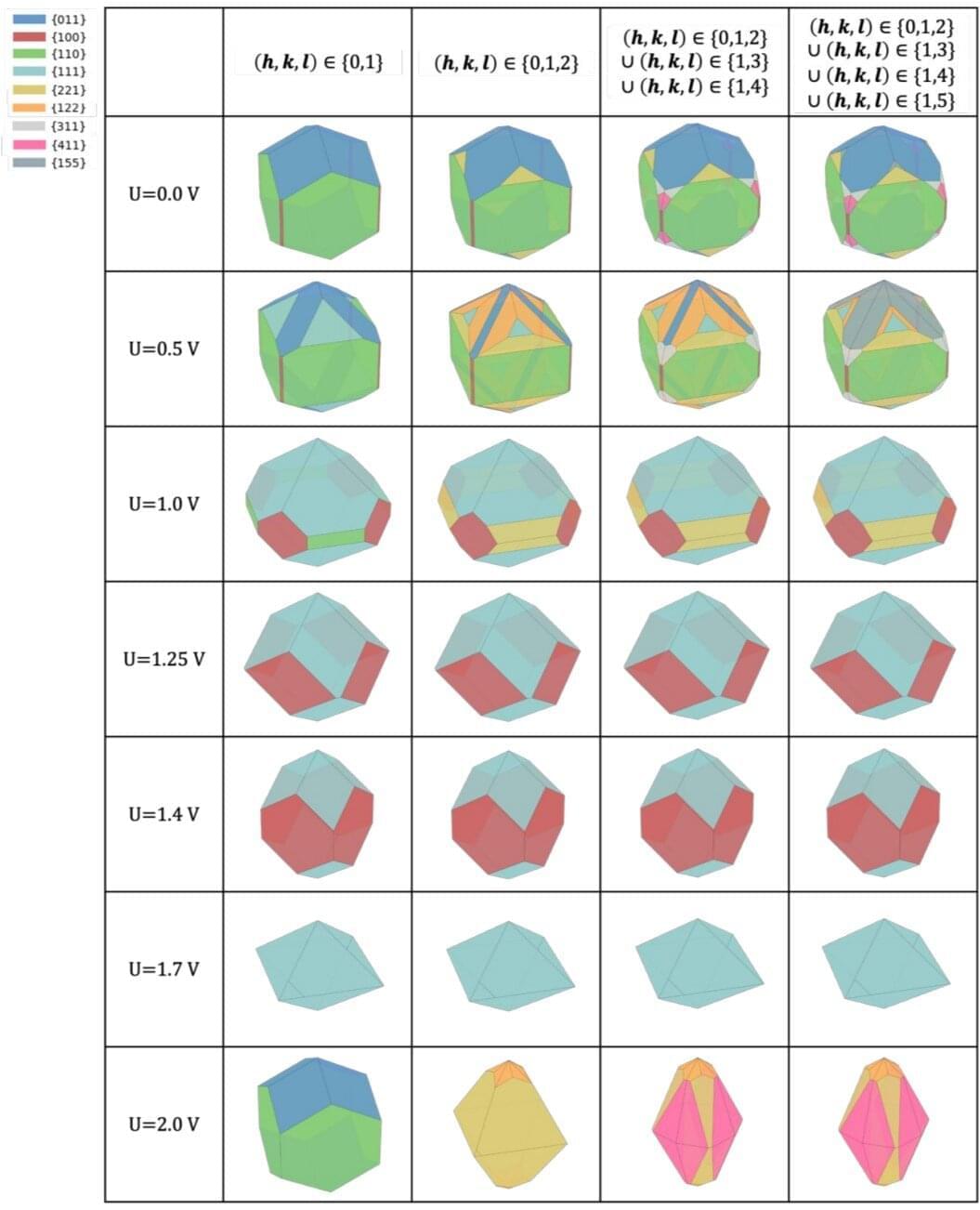
A new study by researchers at Duke University and the University of Pennsylvania offers an unprecedented view of that degradation process, capturing how iridium oxide nanocrystals restructure and dissolve—atom by atom—during electrolysis. The findings provide critical insight into why today’s best catalysts still fail and how future materials might last longer. The study is published in the Journal of the American Chemical Society.