‘The Effect of Exogenous Acid Identity on Iron Tetraphenylporphyrin-Catalyzed CO2 Reduction’ from Inorganic Chemistry is currently free to read as an ACSEditorsChoice.
📖 Read the article.
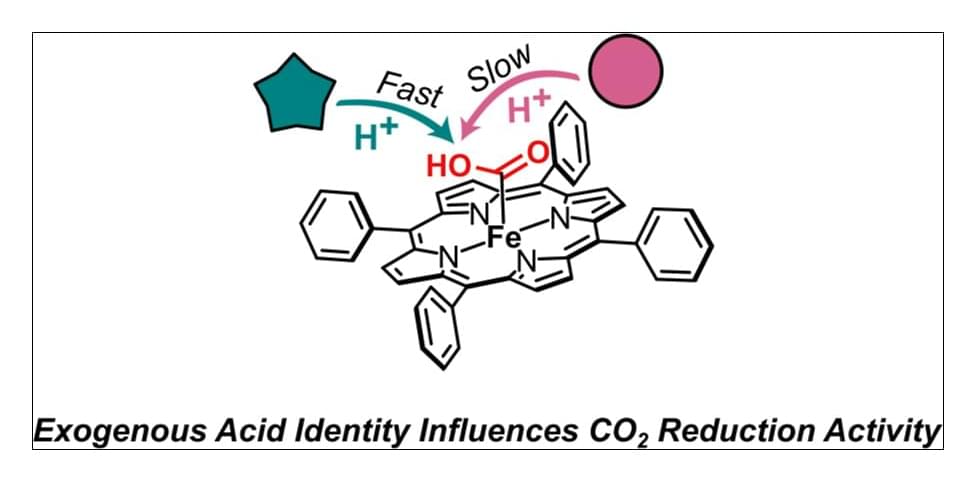
Iron tetraphenylporphyrin (FeTPP) is a privileged electrocatalyst for the 2e–/2H+ reduction of CO2 to CO. FeTPP-catalyzed CO2 reduction typically employs phenol as an exogenous acid to promote the rate-limiting proton-coupled electron transfer. Beyond the observation that catalytic rates increase with decreasing pKa, the effects of acid identity on reaction kinetics are largely unexplored. Herein, we report rates of FeTPP-catalyzed CO2 reduction with structurally diverse O–H, N–H, and C–H acids. While many of these acids follow the expected Brønsted relationship, there are several notable exceptions: the fluorinated alcohols hexafluoroisopropanol (log(kcat) = 4.54) and 2,2,2-trifluoroethanol (log(kcat) = 3.55)─and the N–H acid imidazole (log(kcat) = 4.41)─display catalytic rates that are several times greater than rates obtained with similarly acidic phenols. Amides with pKas 19 (in dimethyl sulfoxide) display similar activity as comparably acidic O–H acids, while rates obtained with less acidic amides are ∼2 orders of magnitude slower than O–H donors of similar pKa. Each C–H acid affords poor activity. An Eyring analysis suggests that acids enforcing less ordered transition states afford superior kinetics. This study reveals that acid pKa is only one relevant parameter for altering catalytic rates, and judicious selection of the acid is crucial for enhancing catalytic rates.