PhD student in Computer Science at Stanford University.
Get the latest international news and world events from around the world.
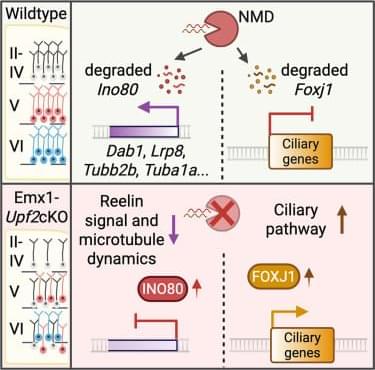
Nonsense-mediated mRNA decay orchestrates neuronal migration and cortical lamination while modulating Reelin and ciliary gene regulatory networks
Lin et al. show that nonsense-mediated mRNA decay (NMD) is essential for neuronal migration and cortical lamination. UPF2 regulates expression of Reelin signaling and microtubule genes via Ino80 and represses ciliary gene Foxj1 to assure normal migration, revealing a key regulated RNA decay mechanism in brain development.

RCC1 depletion drives protein transport defects and rupture in micronuclei
Spotlight: Hiba Baaziz and Daniela Cimini (Virginia Tech) discuss recent work from Zych et al. (https://hubs.la/Q0485YJy0), showing that low RCC1 levels impair protein export in micronuclei, causing overgrowth and rupture. https://hubs.la/Q0485R1g0
Micronuclei (MN), a hallmark of chromosome instability, frequently rupture, leading to protumorigenic consequences. MN rupture requires nuclear lamina defects, yet their underlying causes remain unclear. Here, we demonstrate that MN lamina gaps are linked to excessive MN growth resulting from impaired protein export. This export defect arises from reduced levels of the transport protein RCC1 in MN. Overexpressing RCC1 increases protein export and protects MN from rupture. Differences in RCC1 levels linked to chromatin state also explain why high euchromatin content increases the stability of small MN. Additional RCC1 loss in euchromatic MN results in impaired protein import. For these MN, increasing RCC1, directly or through increasing histone methylation, accelerates rupture. Our findings define a new model of MN rupture, where defects in protein export drives continuous MN growth causing nuclear lamina gaps that predispose MN to membrane rupture and where chromatin-specific features can alter rupture of small MN by further impairing nuclear transport.

Cortically-mediated muscle responses to balance perturbations increase with perturbation magnitude in older adults with and without Parkinson’s disease
New in eNeuro from Boebinger et al: Compared to young adults, older people with and without Parkinson’s disease have larger brain responses and muscle signals that hinder their balance recovery ability.
▶️
We lack a mechanistic understanding of how cortical contributions to balance control change in aging and Parkinson’s disease (PD). Balance is governed by brainstem circuits, with higher-order centers like the cortex or basal ganglia becoming engaged as challenge increases or balance health declines. We previously showed that parallel sensorimotor feedback loops engaging brainstem and cortical circuitry contribute to muscle activity for balance control in young adults (YAs). Here, we analyze data from male and female older adults (OAs) with and without PD, decomposing perturbation-evoked tibialis anterior and medial gastrocnemius muscle activity into hierarchical components based on latencies of feedback control loops. We found that balance-correcting muscle activity followed a stereotypical waveform of long-latency responses (LLRs): LLR1 began ∼120ms and LLR2 occurred ∼210ms, respectively, consistent with subcortical and cortical feedback latencies. Both LLRs increased with balance challenge and could be explained by center of mass kinematics. Perturbation-evoked antagonist muscle activity consisted of destabilizing and stabilizing components categorized based on whether they resist the kinematic errors that drive their activation. The destabilizing component occurred at ∼180ms and was negatively correlated with clinical measures of balance ability in the OA but not PD group. Exploratory comparisons showed OA and PD groups had larger LLR2s at lower challenge levels than YAs, consistent with greater cortical engagement during balance with aging. These findings demonstrate that a neuromechanical model can decompose perturbation-evoked muscle activity into hierarchical components related to clinical balance ability and identify mechanistic changes in the neural control of balance without direct brain measurements.
Significance Statement We show that reactive balance recovery in older adults with and without Parkinson’s disease can be decomposed into distinct components that reflect hierarchical brainstem, cortical, and basal ganglia feedback loops. Using a neuromechanical model of delayed task-level feedback control, we link these components to perturbation difficulty and clinical balance ability in older adults. This framework connects specific features of agonist and antagonist muscle activity to underlying neural control processes without requiring direct brain recordings. Our findings provide a mechanistic basis for age-and disease-related changes in balance control that can inform individualized assessment and future rehabilitation strategies.

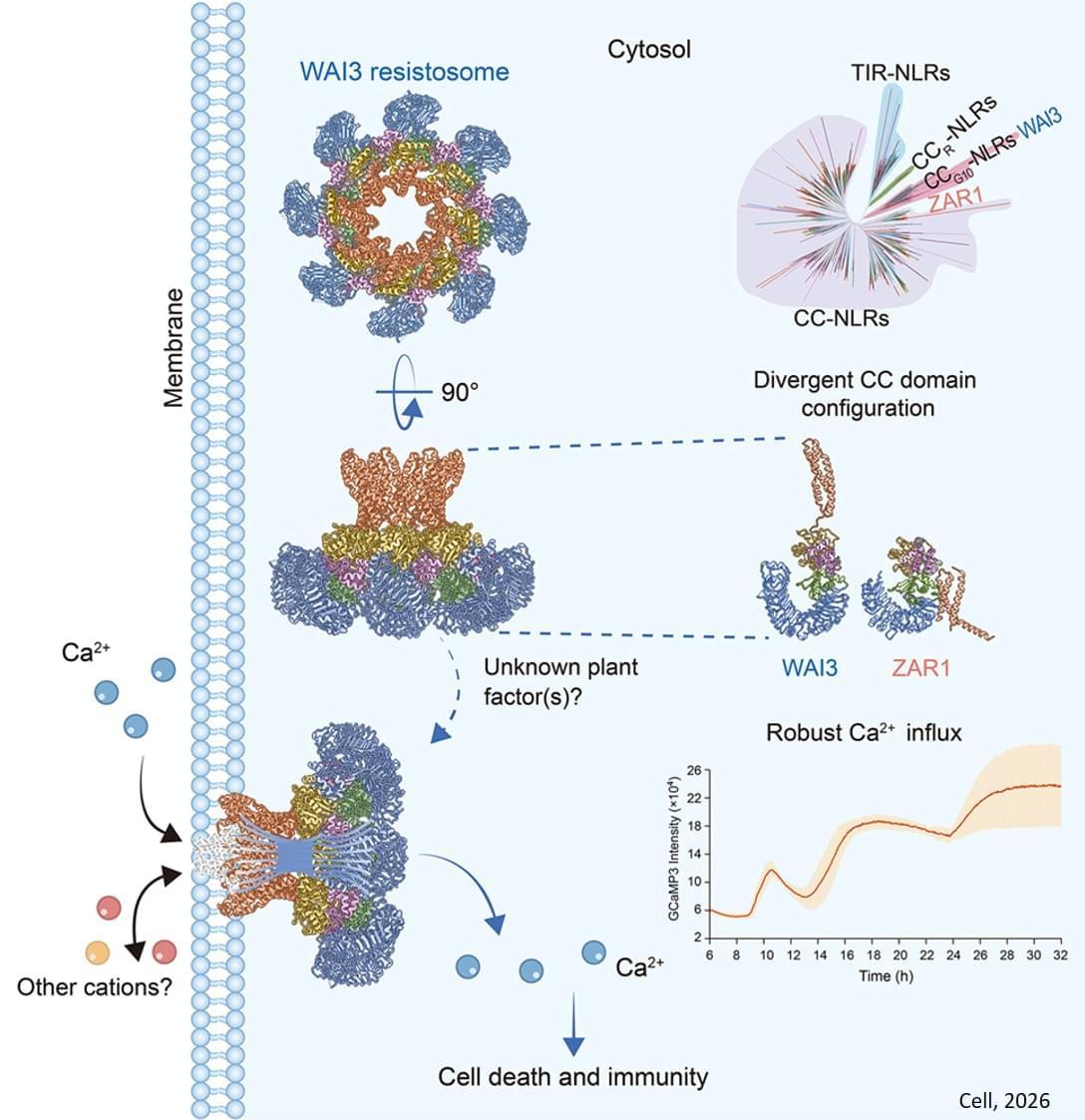
Novel Octameric Resistosome and Immune Mechanism in Wheat
After activation, these NLRs form multi-protein complexes—called resistosomes—that carry out the immune response. Studies have shown that certain resistosomes are pentameric (e.g., ZAR1 and Sr35), whereas others are hexameric (e.g., NRC2 and NRC4). These complexes initiate immune responses by triggering calcium (Ca2+) influx into the cytoplasm. However, the G10 type of CC-NLR (CCG10-NLR) immune receptors constitutes a unique clade among CC-NLRs and its activation mechanism has remained poorly understood.
Now, in a study published in Cell, a research team has revealed a novel octameric resistosome formed by an activated wheat CCG10-NLR immune receptor, which induces Ca2+ influx and immune responses through a unique channel architecture.
The researchers identified the Wheat Autoimmunity 3 (WAI3) gene, which encodes a CCG10-NLR protein. Subsequent analysis revealed that a gain-of-function (GOF) single amino acid mutation in the leucine-rich repeat (LRR) domain leads to autoactivation, providing an opportunity to study the activation mechanism of CCG10-NLR.
After expressing the WAI3 proteins in Nicotiana benthamiana, the researchers used cryo-electron microscopy to resolve the octameric structure of the activated WAI3 resistosome—marking the first time an octameric resistosome has been identified in plants.
The CCG10-NLR WAI3 resistosome differs from known resistosomes both in the number of monomers and in its conformation, representing a novel assembly mechanism for plant NLR resistosomes.
Using Nicotiana benthamiana and animal cell expression systems, the researchers also demonstrated that the WAI3 resistosome induces Ca2+ influx in plants but is not effective in animal cells. ScienceMission sciencenewshighlights.
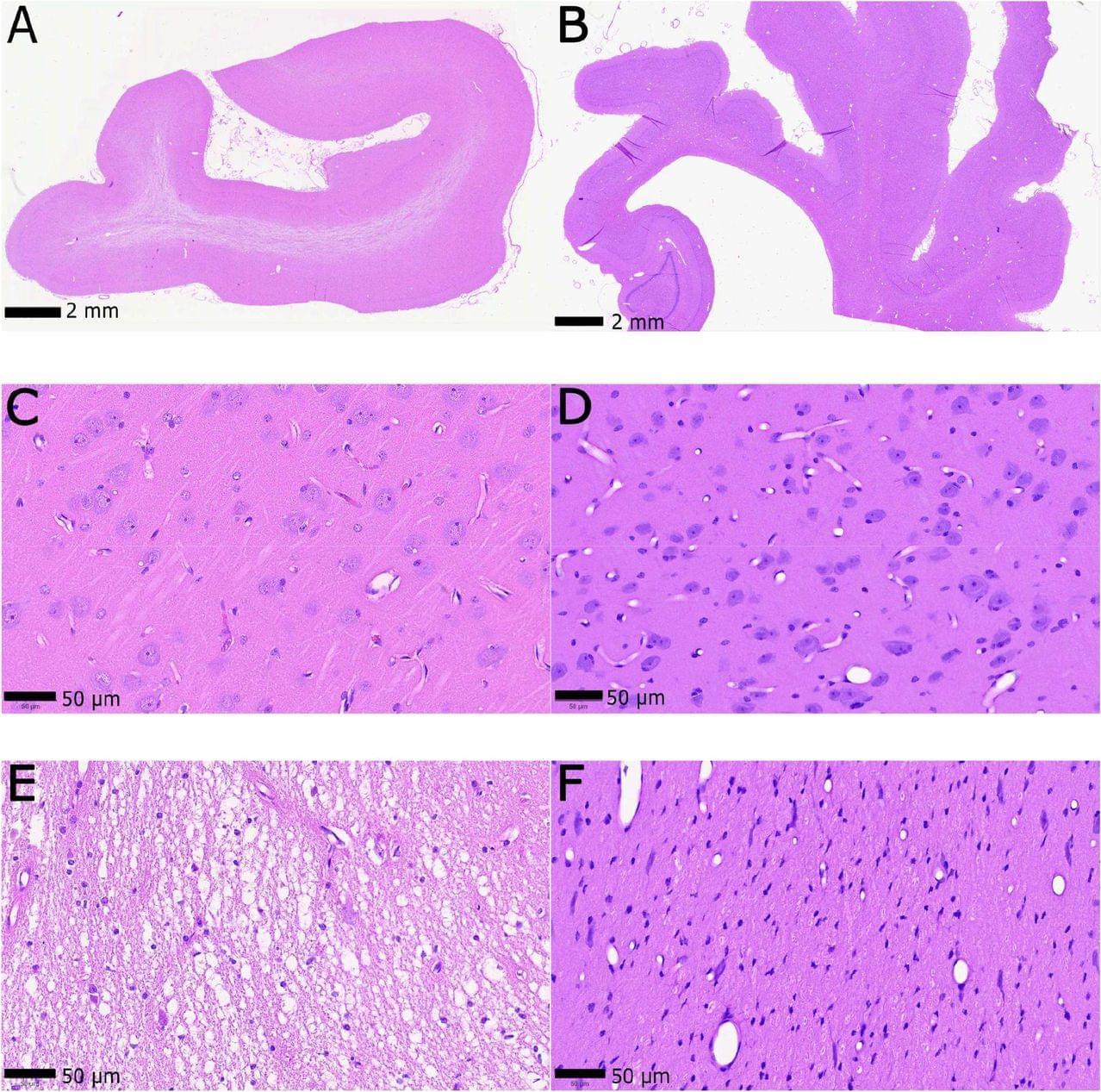
Ultrastructural preservation of a whole large mammal brain with a protocol compatible with human physician-assisted death
Ultrastructural Preservation of a Whole Large Mammal Brain (bioRxiv, 2026) ⚠️ Preprint – not yet peer-reviewed.
A 2026 preprint builds on over a decade of brain preservation research, demonstrating that whole mammalian brains (pigs) can be preserved with remarkable structural fidelity under near–real-world, end-of-life conditions.
The study refines aldehyde-stabilized cryopreservation (ASC)—a technique previously recognized by the Brain Preservation Foundation. This method combines chemical fixation (aldehydes), cryoprotectants, and controlled cooling to prevent ice damage and preserve neural structure at the nanoscale. — What the study shows.
Whole pig brains preserved with intact cellular and synaptic architecture.
Preservation remains viable even with delayed postmortem intervals (~10 minutes)
Tissue remains perfusable and structurally stable after fixation.
Protocol moves toward clinically realistic implementation, not just lab conditions.
The Jellies That Evolved a Different Way To Keep Time
Developmental biologist Tsuyoshi Momose cultures a newly discovered species of jellyfish in a tank of circulating water. Scientists want to understand how these unusual jellies keep time.
The passage of the sun across the sky — dawn, day, dusk, night — drives the clock of life. Some species wake with the sun and sleep with the moon. Others do the opposite, and a few keep odd hours. These naturally driven, 24-hour biological cycles are known as circadian rhythms, and they do more than cue bedtime: They regulate hormones, metabolism, DNA repair, and more. When life falls out of sync, there can be dire consequences for health, reproduction, and survival.
Lacking watches, many species keep time using an internal system — a set of interacting genes and their protein products that effectively keeps track of a 24-hour period — that is calibrated by sunlight. This kind of circadian clock is widespread, found even in single-celled algae, which suggests that biological timekeeping evolved billions of years ago. Across animals, most species have the same genetic system, using genes known as CLOCK, BMAL1, and CRY, or recognizable homologues. This form of biological clock mechanism appears even in ancient lineages, including sponges and some jellyfish.
But is this the only way to do it? In a pea-size jelly off the coast of Japan, biologists are examining a different kind of timekeeping.
NVIDIA CEO Doesn’t Believe in “Succession Plans,” Says He Operates in a Way That Doesn’t Make His Company a One-Man Show
On the Lex Fridman podcast, NVIDIA’s CEO was asked about his mortality and whether he fears dying in his current state. Jensen offered a rather interesting response, saying that his company is currently in the midst of a technological revolution and that, if he died in the meantime, it might not be the best-case scenario for him.
“The most important thing you should do today, if you care about the future of your company, post you, is to pass on knowledge, information, insight, skills, experience as often and continuously as you can. Which is the reason why I continuously reason about everything in front of my team.”
NVIDIA has become the largest business entity and the driving force in the AI world, yet CEO Jensen Huang has no succession plans in sight.