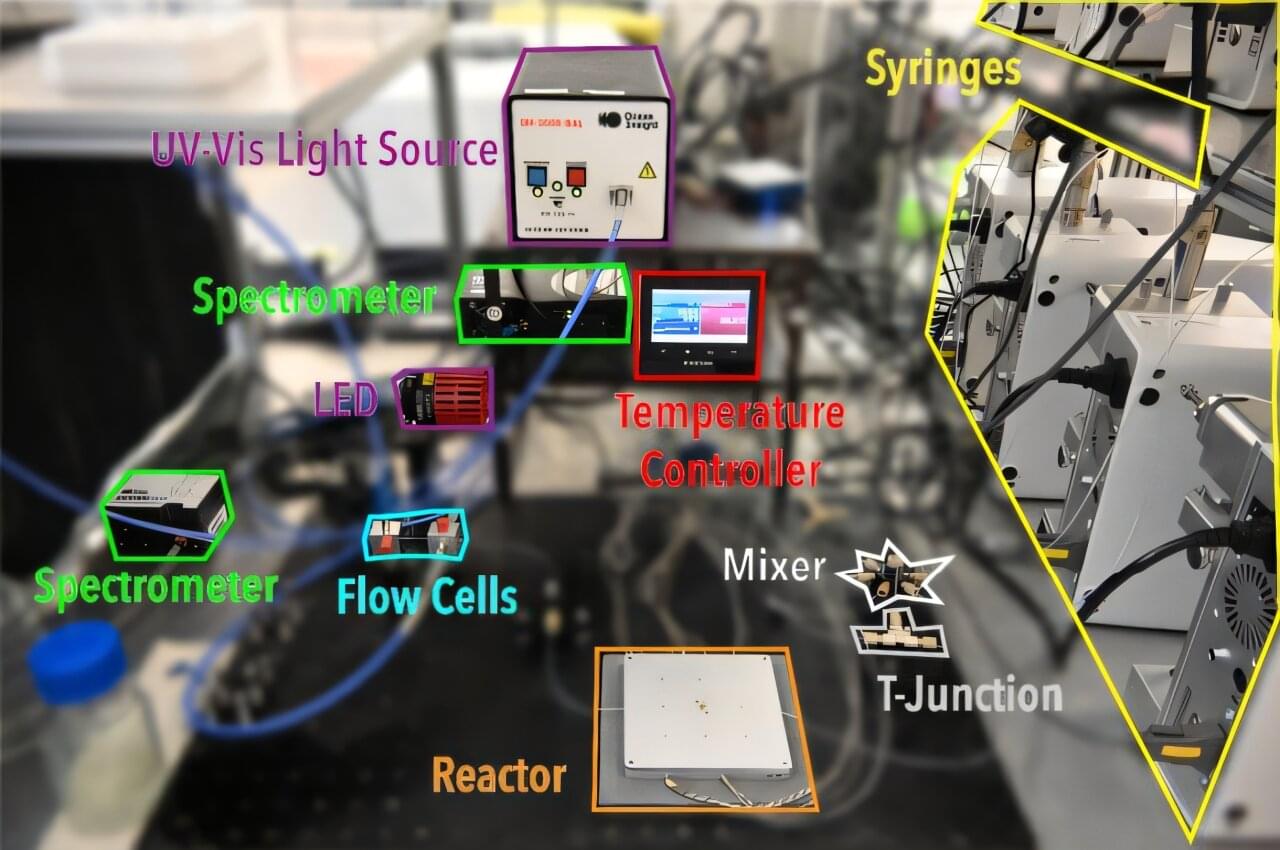
A new autonomous laboratory recently navigated through billions of potential material synthesis recipes to identify brighter, lead-free light-emitting nanomaterials in just 12 hours. The work could accelerate development of safer light-emitting nanoplatelets for use in applications ranging from photodetectors to the production of fuel from solar energy. A paper describing this work appears in Nature Communications.
Nanoplatelets are sheet-like crystals only billionths of a meter thick; in this case, they belong to a family of lead-free “double perovskites,” materials whose atomic recipe can be tuned to control how they absorb and emit light.
“One of the big challenges in developing safer optical nanomaterials is the sheer size of the material universe,” says Milad Abolhasani, Alcoa Professor and University Faculty Scholar in the department of chemical and biomolecular engineering at North Carolina State University. Abolhasani is the corresponding author of the research.