A new study raises important questions about widely used NAD+ supplements, suggesting that compounds often taken to boost energy and support healthy aging may have unintended consequences in cancer treatment.

Researchers at Cornell University have developed a powerful new genetic toolkit that allows scientists to study how genes function at the level of individual cells, an advance that could accelerate discoveries in development, neuroscience and disease.
The system builds on MAGIC (Mosaic Analysis by gRNA-Induced Crossing-over), a method originally created by the labs of Chun Han, associate professor in the Department of Molecular Biology and Genetics in the College of Agriculture and Life Sciences (CALS) and the Weill Institute for Cell and Molecular Biology. MAGIC uses CRISPR gene editing to generate individual mutant cells within otherwise normal tissue, enabling precise comparisons within a living organism.
In the new study, graduate researcher Yifan Shen expanded the approach into a genome-wide toolkit for Drosophila melanogaster, creating resources that work across all chromosomes and allow researchers to study genes that were previously difficult, or impossible, to analyze at single-cell resolution.
Rice behaves in an unexpected way under pressure. When compressed quickly, it becomes weaker, but under slow pressure it stays strong. This insight is helping scientists develop a new material that could be used in “soft” robots that automatically adjust stiffness, as well as protective gear that responds to how fast an impact occurs.
Using this property, researchers created a new type of “metamaterial,” an engineered structure designed to exhibit behaviors not found in natural materials.
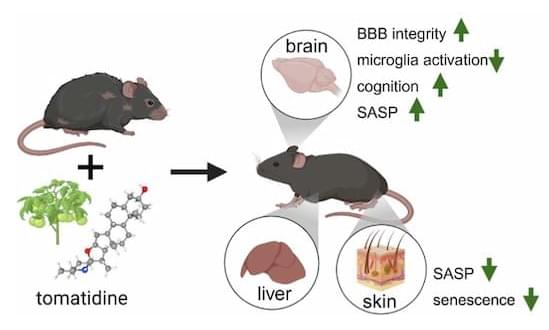
Cellular senescence drives aging and age-related dysfunction across multiple tissues, including the brain. Through a high-content, senescent cell-based phenotypic screen of a small panel of natural products, we identified tomatidine, an aglycone of tomatine found in tomatoes, as a previously unrecognized senotherapeutic agent. In senescent human brain microvascular endothelial cells and fibroblasts, tomatidine selectively suppressed SASP expression without affecting p16Ink4a or p21Cip1 levels consistent with a senomorphic effect. In aged mice, tomatidine reduced frailty and improved motor coordination and cognitive performance. These functional benefits were accompanied by reduced senescence markers (p16 Ink4a, p21 Cip1, and telomere-associated DNA damage foci) in liver, skin, and hippocampal neurons, along with decreased neuroinflammation and microglial activation. Tomatidine also diminished brain endothelial cell senescence while enhancing tight junction protein expression, suggesting preserved blood–brain barrier integrity. Together, these findings identify tomatidine as a promising senescence-targeting compound with beneficial effects in aged mice and support its further evaluation in mechanistic and translational studies.
We report that the RNA-editing enzyme ADAR1 downregulates nuclear-and mitochondria-encoded double-stranded RNAs (dsRNAs) to maintain immune homeostasis. ADAR1 employs RNA-editing-dependent and-independent mechanisms to keep dsRNA levels low in cells. Notably, upon ADAR1 loss, mitochondrial dsRNA levels increase and can cause enhanced inflammation upon mitochondrial stress.
The new observations indicate that it was a very peculiar comet… up to a point.