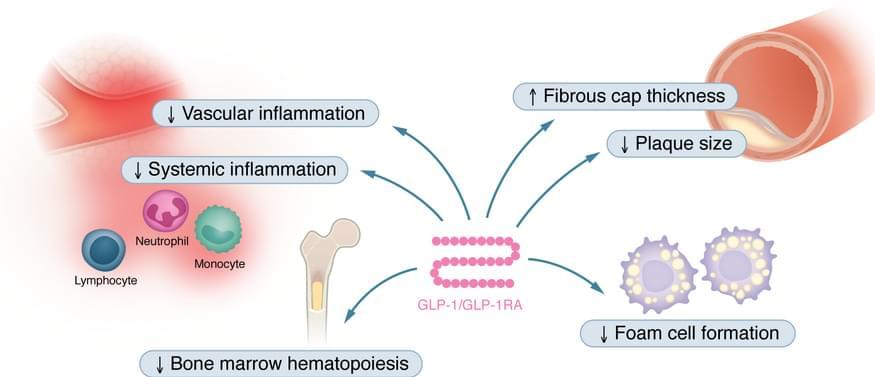
As part of JCI’s Review Series on Clinical Innovation and Scientific Progress in GLP-1 Medicine Florian Kahles, Andreas L. Birkenfeld, & Nikolaus Marx summarize the effects of GLP-1 and GLP-1RAs in the cardiovascular system as well as clinical data of GLP-1RAs in individuals with cardiovascular disease or in those at high risk.
1Department of Internal Medicine I, University Hospital Aachen, RWTH Aachen, Aachen, Germany.
2German Center for Diabetes Research (DZD), Neuherberg, Germany.
3Department of Internal Medicine IV, Diabetology, Endocrinology and Nephrology, Eberhard-Karls University Tübingen, Tübingen, Germany.