Novel study by researchers at the Rambam Health Care Campus could help 21 million people worldwide suffering from the severe lifelong mental disorder


“Working in strict secrecy, a government scientist in Norway built a machine capable of emitting powerful pulses of microwave energy and, in an effort to prove such devices are harmless to humans, in 2024 tested it on himself. He suffered neurological symptoms similar to those of ”Havana syndrome,” the unexplained malady that has struck hundreds of U.S. spies and diplomats around the world.
The bizarre story, described by four people familiar with the events, is the latest wrinkle in the decade-long quest to find the causes of Havana syndrome, whose sufferers experience long-lasting effects including cognitive challenges, dizziness and nausea. The U.S. government calls the events Anomalous Health Incidents (AHIs).
The secret test in Norway has not been previously reported. The Norwegian government told the CIA about the results, two of the people said, prompting at least two visits in 2024 to Norway by Pentagon and White House officials.
The CIA investigated a Norwegian government experiment with a pulsed-energy machine in which a researcher built and tested a ”Havana syndrome” device on himself.
When HHMI Investigator Amita Sehgal started studying sleep 25 years ago, the topic elicited a yawn from most biologists. “In the year 2000, if I had suggested to my department that we hire people working on sleep, they would have laughed at me,” says Sehgal, a molecular biologist and neuroscientist at the University of Pennsylvania. “The thinking was that sleep is not something that neuroscientists do; psychologists study sleep and dreams.” Now, more than two decades later, sleep science has finally woken up.
Biologists around the world are now studying sleep in everything from fruit flies to jellyfish to understand the fundamental molecular and cellular mechanisms that drive slumber and answer the age-old question of why we sleep.
“Sleep is widely conserved across the animal kingdom and so it must have some basic function that is the same across species, and so what is that?” Sehgal says. “We’re finally getting to a point where we are recognizing a few basic principles about sleep.”

Researchers at Washington University School of Medicine in St. Louis have developed a method to predict when someone is likely to develop symptoms of Alzheimer’s disease using a single blood test. In a study published in Nature Medicine, the researchers demonstrated that their models predicted the onset of Alzheimer’s symptoms within a margin of three to four years.
This method could have implications both for clinical trials developing preventive Alzheimer’s treatments and for eventually identifying individuals likely to benefit from these treatments.
More than seven million Americans live with Alzheimer’s disease, with health and long-term care costs for Alzheimer’s and other forms of dementia projected to reach nearly $400 billion in 2025, according to the Alzheimer’s Association. This massive public health burden currently has no cure, but predictive models could help efforts to develop treatments that prevent or slow the onset of Alzheimer’s symptoms.
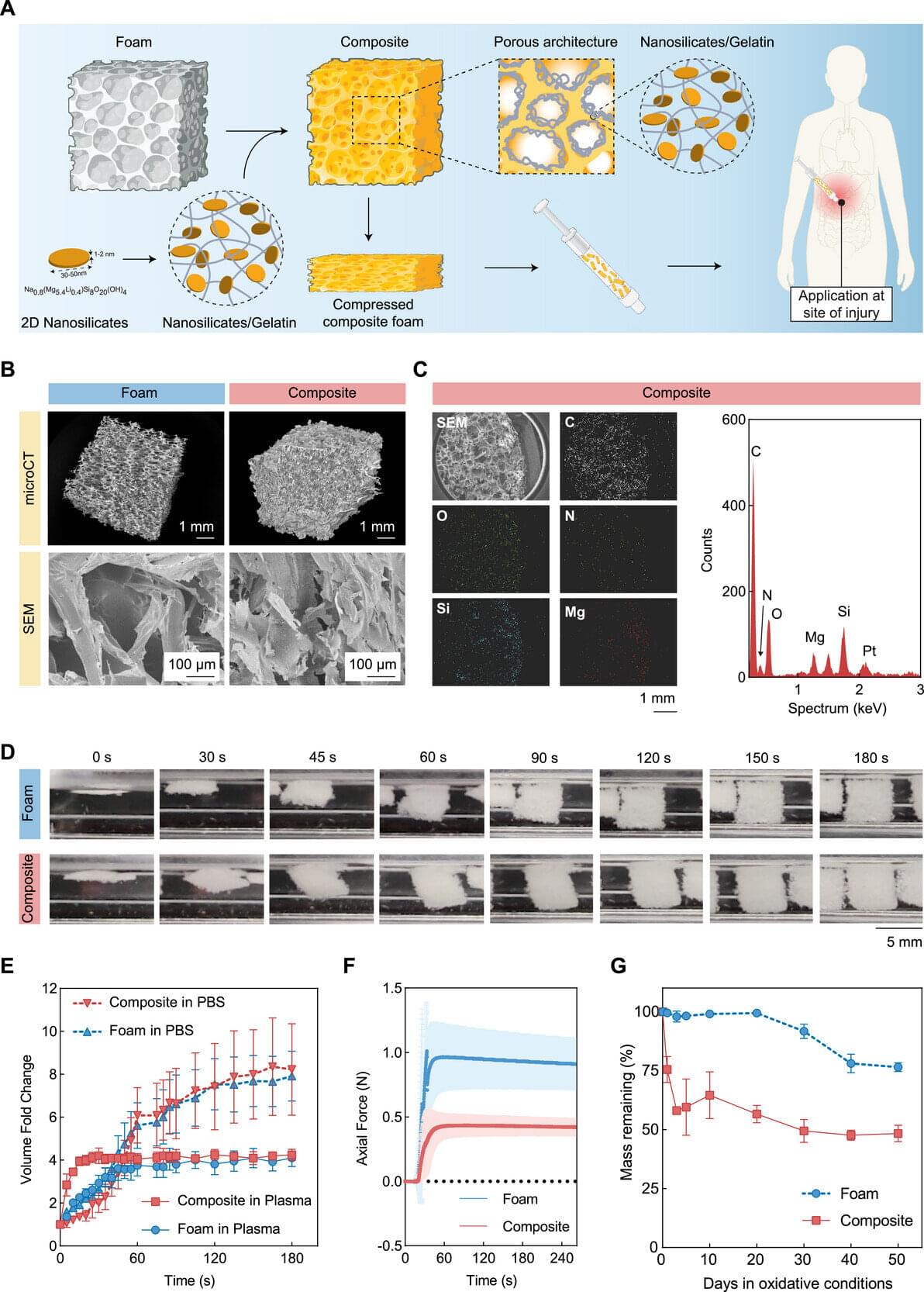
Traumatic injury is the third leading cause of death in the state of Texas, surpassing strokes, Alzheimer’s disease and diabetes, according to the Centers for Disease Control and Prevention. A massive number of these deaths are the result of uncontrolled bleeding. “Severe blood loss can rapidly lead to hemorrhagic shock,” said Dr. Akhilesh Gaharwar, a biomedical engineering professor at Texas A&M University. “Many patients die within one to two hours of injury. This critical period is often referred to as the ‘golden hour.’”
Gaharwar and his fellow researchers in the biomedical engineering department have found a way to extend this golden hour—using clay.
Gaharwar, Dr. Duncan Maitland and Dr. Taylor Ware are developing a suite of injectable hemostatic bandages —biomedical materials that stop bleeding and promote blood to clot faster. Their research is specifically targeting deep internal bleeding where traditional methods like compression are not possible.
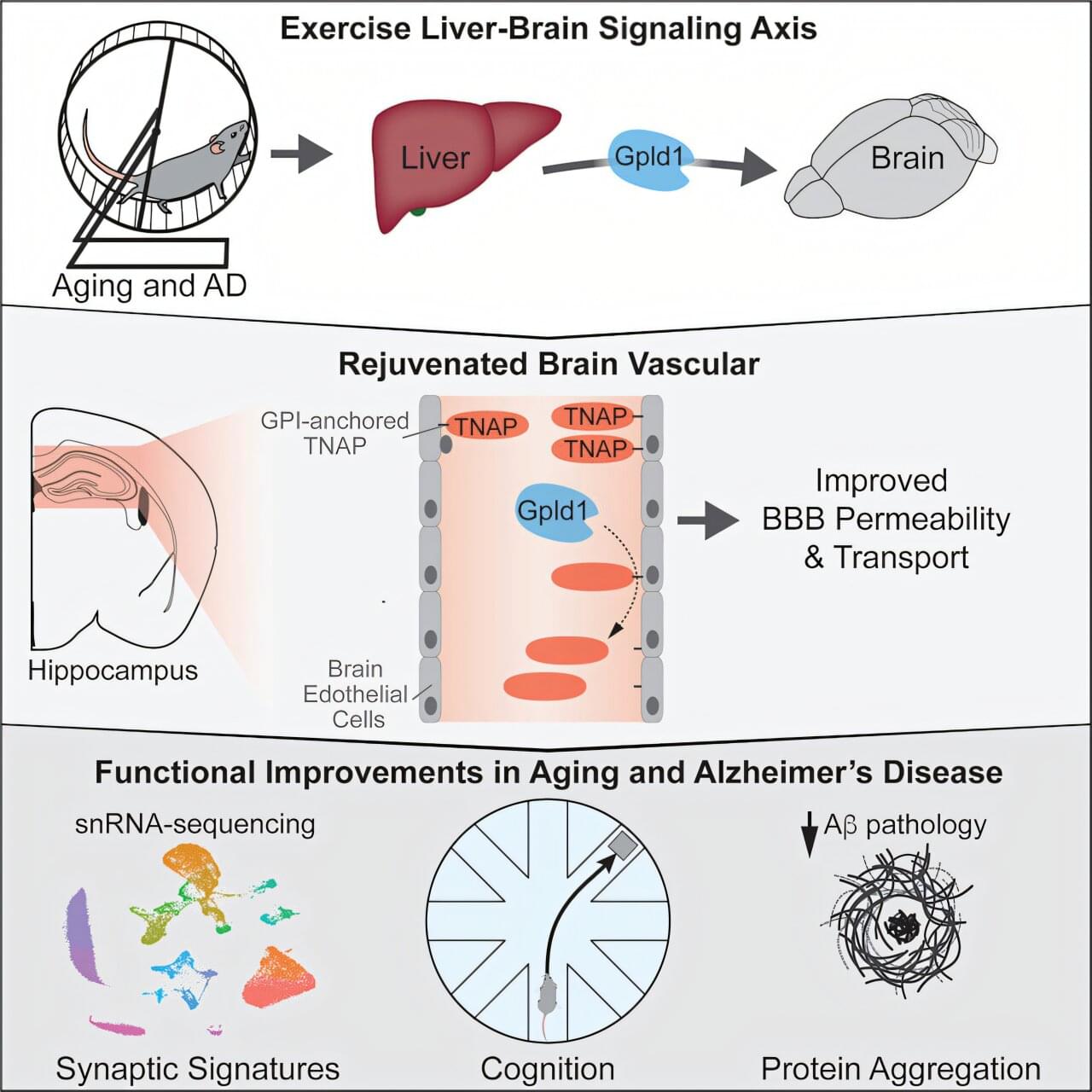
Researchers at UC San Francisco have discovered a mechanism that could explain how exercise improves cognition by shoring up the brain’s protective barrier. With age, the network of blood vessels—called the blood–brain barrier—gets leaky, letting harmful compounds enter the brain. This causes inflammation, which is associated with cognitive decline and is seen in conditions like Alzheimer’s disease. The research is published in the journal Cell.
Six years ago, the team identified a brain-rejuvenating enzyme called GPLD1 that mice produced in their livers when they exercised. But they couldn’t understand how it worked, because it cannot get into the brain.
The new study answers that question. Researchers discovered that GPLD1 was working through another protein called TNAP. As the mice age, the cells that form the blood-brain barrier accumulate TNAP, which makes it leaky. But when mice exercise, their livers produce GPLD1. It travels to the vessels that surround the brain and trims TNAP off the cells.

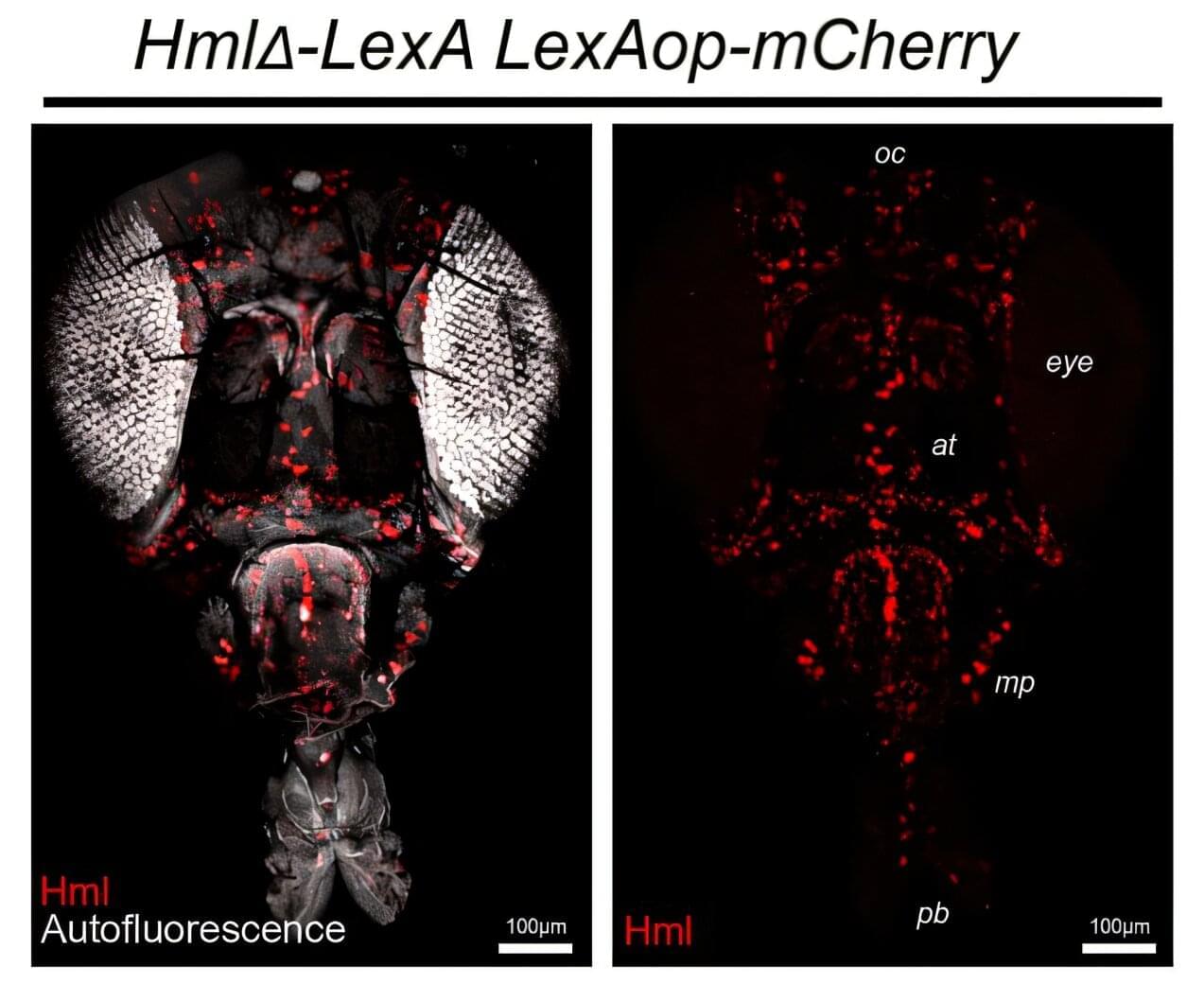
A team led by Northwestern University and Shirley Ryan AbilityLab scientists have developed a new technology that can eavesdrop on the hidden electrical dialogues unfolding inside miniature, lab-grown human brain-like tissues. Known as human neural organoids—and sometimes called “mini brains”—these millimeter-sized structures are powerful models of brain development and disease. But until now, scientists could only record and stimulate activity from a small fraction of their neurons—missing network-wide dynamics that give rise to coordinated rhythms, information processing and the complex patterns of activity that define brain function.
For the first time, the new technology overcomes that stubborn limitation. The soft, three-dimensional (3D) electronic framework wraps around an organoid like a breathable, high-tech mesh. Rather than sampling select regions, it delivers near-complete, shape-conforming coverage with hundreds of miniaturized electrodes. That dense, three-dimensional interfacing enables scientists to map and manipulate neural activity across almost the entire organoid.
By moving from localized probing to true whole-network mapping, the work brings organoid research closer to capturing how real human brains develop, function and even fail.
In a large US-based brain imaging study, researchers found that these drugs do not primarily affect attention networks, but instead act on systems linked to arousal, sleep, and motivation.
The puzzle of ADHD stimulants
Prescription stimulants such as methylphenidate and amphetamines are among the most used psychoactive drugs in children and adolescents with ADHD, where they remain a first-line treatment. Estimates for receiving a prescription for ADHD medication among diagnosed children vary from 38–81%. Despite their widespread use, there is still disagreement about how these drugs work in the brain.

Tumor-immune-neural circuit in cancer cachexia.
The mechanisms involved in cancer-mediated cachexia and anorexia are not well understood.
The researchers in this study delineate an interplay among tumor cells, immune cells, and the nervous system that drives cancer cachexia and anorexia.
The authors show thay loss of GDF15 protects against appetite loss, muscle wasting, and fat loss in pancreatic, lung, and skin cancers.
Disrupting this feedforward loop with GDF15-neutralizing antibody, anti-CSF1R antibody, or Rearranged during Transfection (RET) inhibitor alleviates cachexia and anorexia across cancer models. sciencenewshighlights ScienceMission https://sciencemission.com/Tumor-immune-neural-circuit
Shi et al. delineate an interplay among tumor cells, immune cells, and the nervous system that drives cancer cachexia and anorexia. Specifically, tumor-derived CSF1 induces macrophage GDF15, which signals through the GFRAL-RET neural axis to enhance β-adrenergic activity and systemic wasting. Disrupting this feedforward loop alleviates cachexia across cancer models.
