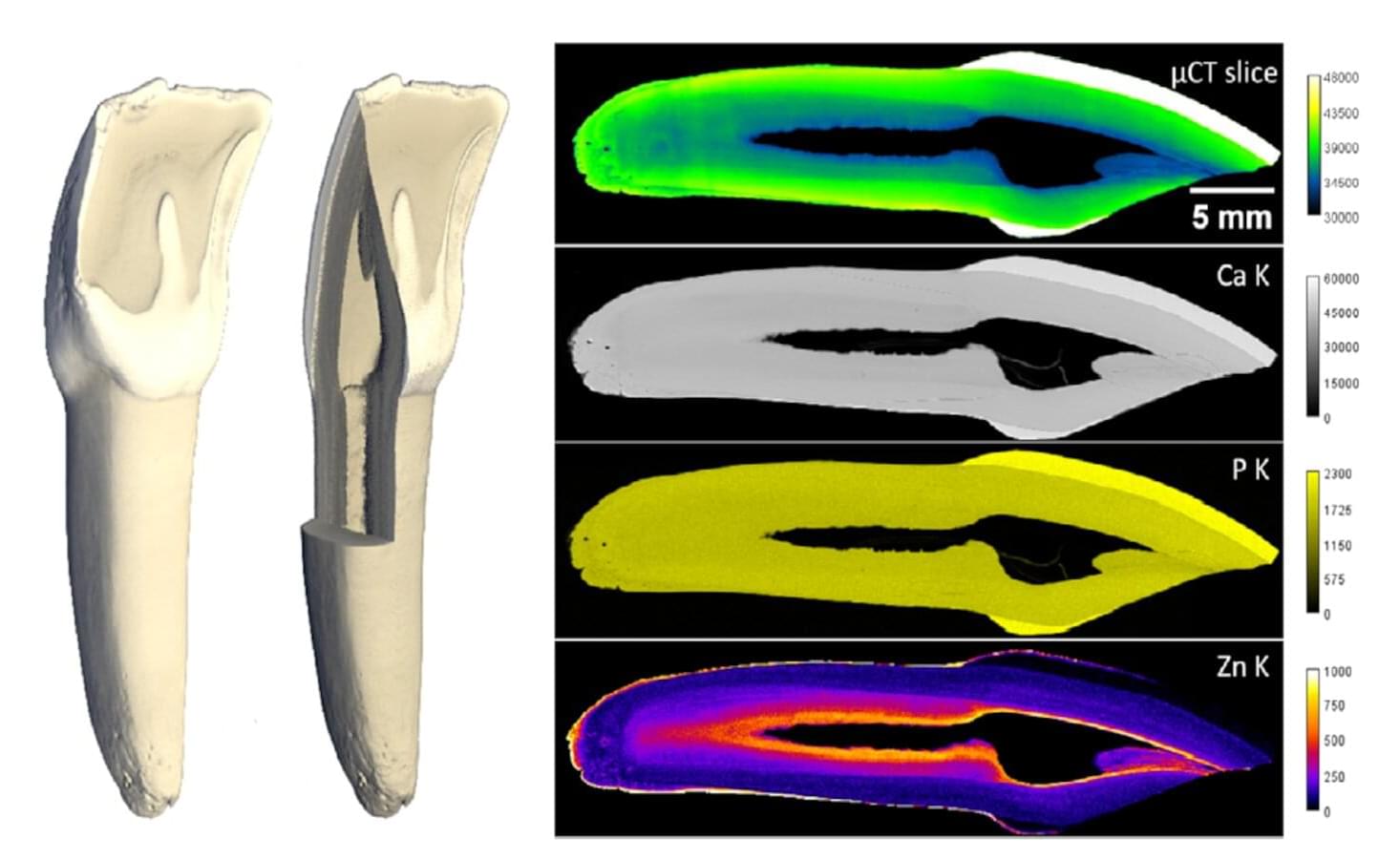
Teeth are composites of mineral and protein, with a bulk of bony dentin that is highly porous. This structure allows teeth to be both strong and sensitive. Besides calcium and phosphate, teeth contain trace elements such as zinc. Using complementary microscopy imaging techniques, a team from Charité Berlin, TU Berlin and HZB has quantified the distribution of natural zinc along and across teeth in 3 dimensions. The team found that, as porosity in dentin increases towards the pulp, zinc concentration increases 5~10 fold. These results help to understand the influence of widely-used zinc-containing biomaterials (e.g. filling) and could inspire improvements in dental medicine.
The paper is published in the journal VIEW.
Teeth have a complex structure: the dental pulp with the nerves is surrounded by dentin, a porous bony material, covered externally by enamel in the mouth and cementum in the roots. Although dentin is criss-crossed by countless micrometer-sized dentin tubules, teeth can withstand decades of cyclic, repeated forces. The density of the dentinal tubules increases towards the pulp, meaning that the dentin becomes increasingly porous towards the inside.